Health Technology Assessment (HTA)
&
Evidence-informed Health Policymaking (EIHP)
&
Evidence-informed Health Policymaking (EIHP)
Ministry of Health and Care Services informed the Parliament (Stortinget) (Meld. St. 10 (2012–2013) of the January 2013 launch of: "The National system for the introduction of new health technologies within the specialist health service – For better and safer patient care" ("Nasjonalt system for innføring av nye metoder i spesialisthelsetjenesten – for bedre og tryggere pasientbehandling") with Directorate of Health assuming the secretariat functions and responsibilities.
Interestingly, the January 2013 launch of this "National system for..." was 22 months, almost 2 years, prior to the implementation of Directorate of Health's knowledge-obviated, science-bending, medically & ethically flawed 2014 Recommendation with their exclusive implementation of NCFM eSnurra Group's "method" (i.e., the appropriated, plagiarized, misused Hutchon Method of PDEE) within a government-mandated protocol of evidence-obviated medicine; a protocol proven to cause increased medical risks, critical medical mistakes and grievous medicals harms.
However, and strangely, Ministry of Health and Care Services' new system, "The National system for the introduction of new health technologies within the specialist health service – For better and safer patient care", which was given the shorter name of: New Methods System (Nye Metoder System), was bypassed, completely, for Directorate of Health's national 2014 Recommendation without any explanation, whatsoever. Remember, Directorate of Health's 2014 Recommendation resulted from a science-bending comparison of two pregnancy dating methods: 1) Bergen Group's Terminhjulet method and NCFM eSnurra Group's eSnurra "method," each of which predicts/estimates that which the other does not predict/estimate for objectives which are unique to each method. Yes, you read that correctly; because claiming these two methods are comparable is no small part of the science-bending involved.
Also, Norwegian Directorate of Health had been the secretariat of the National System for the introduction of new methods in the specialist health service, New Methods. The secretariat is responsible for the preparation and follow-up of decisions in the Ordering Forum RHF, and operates the website www.nyemetoder.no. Moreover, he Secretariat participates in the EU's Joint Action on relevant areas such as evidence generation as well as national implementation and reuse of method assessments produced in European cooperation.
Health Technology Assessment (HTA)
The Norwegian Directorate of Health (Helsedirectoratet) defines Health Technology Assessment (HTA) as follows.
Health Technology Assessment Ignored
The Health Technology Assessment (HTA) process, which has been in place in Norway for the past 20 years, was ignored by: Ministry of Health and Care Services, Directorate of Health, NCFM eSnurra Group and NOKC with respect to Directorate of Health's knowledge-obviated, medically & ethically flawed 2014 Recommendation (i.e., national medical policy) with their exclusive implementation of NCFM eSnurra Group's "method" (i.e., the appropriated, plagiarized, misused Hutchon Method of PDEE) within a government-mandated protocol of evidence-obviated medicine; a protocol proven to cause increased medical risks, critical medical mistakes and grievous medical harms with respect to obstetric medicine, fetal medicine and obstetric clinical care. Included below are relevant excerpts from "The National system for the introduction of new health technologies within the specialist health service – For better and safer patient care" Norwegian version: "Nasjonalt system for innføring av nye metoder i spesialisthelsetjenesten – for bedre og tryggere pasientbehandling"
- "Fra 2012 innføres det et system for nye og kostnadskrevende behandlingsmetoder i spesialisthelsetjenesten. Systemet innebærer at det skal gjennomføres metodevurdering før det tas beslutninger om å innføre nye metoder. Systemet skal bidra til større åpenhet i beslutningsprosessen og mer kunnskapsbaserte beslutninger. Se nærmere omtale i Kapittel 8 Mer systematisk utprøving og innføring av nye behandlingsmetoder."
"From 2012, a system will be introduced for new and costly treatment methods in the specialist health service. The system entails that methodological assessment is carried out before decisions are taken to introduce new methods. The system will contribute to greater openness in the decision-making process and more knowledge-based decisions. See Chapter 8, more systematic testing and introduction of new treatment methods." (Source: Meld. St. 10 (2012–2013) God kvalitet – trygge tjenester— Kvalitet og pasientsikkerhet i helse- og omsorgstjenesten, Chapter 1 Introduction)
Interestingly, the January 2013 launch of this "National system for..." was 22 months, almost 2 years, prior to the implementation of Directorate of Health's knowledge-obviated, science-bending, medically & ethically flawed 2014 Recommendation with their exclusive implementation of NCFM eSnurra Group's "method" (i.e., the appropriated, plagiarized, misused Hutchon Method of PDEE) within a government-mandated protocol of evidence-obviated medicine; a protocol proven to cause increased medical risks, critical medical mistakes and grievous medicals harms.
However, and strangely, Ministry of Health and Care Services' new system, "The National system for the introduction of new health technologies within the specialist health service – For better and safer patient care", which was given the shorter name of: New Methods System (Nye Metoder System), was bypassed, completely, for Directorate of Health's national 2014 Recommendation without any explanation, whatsoever. Remember, Directorate of Health's 2014 Recommendation resulted from a science-bending comparison of two pregnancy dating methods: 1) Bergen Group's Terminhjulet method and NCFM eSnurra Group's eSnurra "method," each of which predicts/estimates that which the other does not predict/estimate for objectives which are unique to each method. Yes, you read that correctly; because claiming these two methods are comparable is no small part of the science-bending involved.
- "We hold the opinion that the comparison conducted by the Trondheim Group suffers from conflicts of interest, but the Directorate of Health appears not to have realized that the comparative evaluation of the two methods really amounts to comparing apples to pears." (Source: "Flawed recommendation issued by the Norwegian Directorate of Health concerning the determination of fetal age" or "Helsedirektoratet gir feil anbefaling om bestemmelse av fosteralder" Cathrine Ebbing, MD, PhD, Synnøve Lian Johnsen MD, PhD, Jørg Kessler, MD, PhD, Torvid Kiserud, MD, PhD, Svein Rasmussen, MD, PhD., Nr. 8, 5 mai 2015, Tidsskr Nor Legeforen, 2015; 135:7401, DOI: 10.4045/tidsskr.15.0. HTML versions:English or Norwegian)
Also, Norwegian Directorate of Health had been the secretariat of the National System for the introduction of new methods in the specialist health service, New Methods. The secretariat is responsible for the preparation and follow-up of decisions in the Ordering Forum RHF, and operates the website www.nyemetoder.no. Moreover, he Secretariat participates in the EU's Joint Action on relevant areas such as evidence generation as well as national implementation and reuse of method assessments produced in European cooperation.
- Following the decision of the HOD from May 2017, the Secretariat for New Methods will be transferred to the regional health authorities of Helse Sør Øst RHF. The relocation of the Secretariat to Health South-East is implemented from 1 January 2018."
"Etter beslutning av HOD fra mai 2017 overføres Sekretariatet for Nye metoder til de regionale helseforetakene ved Helse Sør-Øst RHF. Flyttingen av Sekretariatet til Helse Sør-Øst iverksettes fra 1. januar 2018." (Source: HELSEDIREKTORATET – ÅRSRAPPORT 2017, p. 49)
- 19.4 National system for introducing new methods in the specialist health service
19.4 Nasjonalt system for innføring av nye metoder i spesialisthelsetjenesten
A new system for introducing new and costly methods in the specialist health service is described in Meld. St. 16 (2010-2011) National Health and Care Plan (2011-2015) and in Meld. St. 10 (2012-2013) Good quality - safe services.
Et nytt system for innføring av nye og kostnadskrevende metoder i spesialisthelsetjenesten er beskrevet i Meld. St. 16 (2010–2011) nasjonal helse- og omsorgsplan (2011–2015) og i Meld. St. 10 (2012–2013) God kvalitet – trygge tjenester.
The system was established in 2012 and launched in January 2013. A secretariat has been established in the Directorate of Health, a national working group with all the key actors, a reference group with representatives from users and industry, as well as the Order Forum RHF. A new Decision Forum for new methods was established in May 2014.
Systemet ble etablert i 2012 og lansert i januar 2013. Det er opprettet et sekretariat i Helsedirektoratet, en nasjonal arbeidsgruppe med alle de sentrale aktørene, en referansegruppe med representanter fra brukere og industrien, samt Bestillerforum
RHF. Beslutningsforum for nye metoder ble etablert i mai 2014.
The purpose of the system is to increase quality and patient safety in treatment services by helping patients access new, effective methods as quickly as possible, and that ineffective or harmful treatment methods are not used. For medicines that have a marketing authorization and thus have a proven positive benefit / risk ratio, the system will help to promote an economically and medicinally correct use of the drugs in the specialist health service.
Formålet med systemet er å øke kvalitet og pasientsikkerhet i behandlingstilbudet ved å bidra til at pasienter så raskt som mulig får tilgang til nye, virkningsfulle metoder og at behandlingsmetoder som er ineffektive eller skadelige ikke brukes. For legemidler som har markedsføringstillatelse og dermed har et dokumentert positivt nytte/risikoforhold, vil systemet bidra til å fremme en økonomisk og medisinsk riktig bruk av legemidlene i spesialisthelsetjenesten.
The system employs internationally recognized standards for methodological assessment (HTA) that ensure that decisions are based on the best possible academic knowledge base. Method assessment is a systematic summary and assessment of available scientific literature. The assessments may include medical devices and apparatus for prevention, investigation / diagnosis, treatment and follow-up of procedures, medicines, care, as well as organization of service and other interventions.
Systemet tar i bruk internasjonalt anerkjente standarder for metodevurdering (Health Technology Assessment – HTA) som sikrer at beslutningene er basert på et best mulig faglig kunnskapsgrunnlag. Metodevurdering er en systematisk oppsummering og vurdering av tilgjengelig vitenskapelig litteratur. Vurderingene kan omfatte medisinsk utstyr og apparatur til forebygging, utredning/diagnostikk, behandling og oppfølging av prosedyrer, legemidler, pleie, samt organisering av tjenesten og andre intervensjoner. - 19.4.1 Structure
19.4.1 Struktur
The system is based on the principles of openness and transparency and is implemented and developed within today's governance structures and legislation.
Systemet bygger på prinsippene om åpenhet og transparens og implementeres og utvikles innenfor dagens styringsstrukturer og lovverk.
(Source: "Meld. St. 28 (2014–2015) Melding til Stortinget, Legemiddelmeldingen, Riktig bruk – bedre helse" (Meld. St. 28 (2014-2015) Message to the Storting, Drug Report, Proper Use - Better Health. 19.4 Nasjonalt system for innføring av nye metoder i spesialisthelsetjenesten, p. 124-127)
Health Technology Assessment (HTA)
The Norwegian Directorate of Health (Helsedirectoratet) defines Health Technology Assessment (HTA) as follows.
- "HTA Health Technology Assessment. Tools that will help to make knowledge-based decisions when introducing new methods in the health service. Should elucidate the effects and safety-related, health-economic, ethical, legal and social consequences of the method"
"HTA Health Technology Assessment. Vektøy som skal bidra til å ta kunnskapsbaserte beslutninger ved innføring av nye metoder i helsetjenesten. Bør belyse effekt og sikkerhetsmessige, helseøkonomiske, etiske, juridiske og sosiale konsekvenser knyttet til metoden" (Source: "Guide for the development of knowledge-based guidelines" ("Veileder for utvikling av kunnskapsbaserte retningslinjer") Redaktør: Caroline Hodt-Billington. Utgitt: 10/2012. Publikasjonsnummer: IS-1870. ISBN-nr. 978-82-8081-225-4. Utgitt av: Helsedirektoratet, 2012. p. 7) - View/Download Norwegian Directorate of Health's 2-page "Checklist for the development of knowledge-based guidelines" (Sjekkliste for utvikling av kunnskapsbaserte retningslinjer) View/Download Norwegian Directorate of Health's 58-page "Guide for the development of knowledge-based guidelines" (Veileder for utvikling av kunnskapsbaserte retningslinjer) (Source: Helsedirectoratet website: https://www.helsedirektoratet.no/veiledere/utvikling-av-kunnskapsbaserte-retningslinjer)
Health Technology Assessment Ignored
The Health Technology Assessment (HTA) process, which has been in place in Norway for the past 20 years, was ignored by: Ministry of Health and Care Services, Directorate of Health, NCFM eSnurra Group and NOKC with respect to Directorate of Health's knowledge-obviated, medically & ethically flawed 2014 Recommendation (i.e., national medical policy) with their exclusive implementation of NCFM eSnurra Group's "method" (i.e., the appropriated, plagiarized, misused Hutchon Method of PDEE) within a government-mandated protocol of evidence-obviated medicine; a protocol proven to cause increased medical risks, critical medical mistakes and grievous medical harms with respect to obstetric medicine, fetal medicine and obstetric clinical care. Included below are relevant excerpts from "The National system for the introduction of new health technologies within the specialist health service – For better and safer patient care" Norwegian version: "Nasjonalt system for innføring av nye metoder i spesialisthelsetjenesten – for bedre og tryggere pasientbehandling"
- "5.0 Introduction
New health technologies can offer opportunities for health benefits, but can also present challenges linked to prioritisation and resource use. Ever stricter requirements are being imposed on the assessment of new health technologies to ensure patient safety in connection to their introduction and use. To date, there has been no provision for the consistent and systematic assessment of new health technologies within the specialist health service. For the purposes of this document, the term ‘health technology’ means all measures that are used to prevent, investigate, diagnose and treat diseases 3, as well as measures for the rehabilitation of patients and the organisation of health services. [3 Including somatic care, psychiatry and substance abuse]
A national system for the introduction of new health technologies in the specialist health service is implemented. The key aspects of this new system are described in “Nasjonal helse- og omsorgsplan 2011-2015” [3] and “Stortingsmelding 10 (2012-2013), God kvalitet – trygge tjenester” [4]. This document presents a more detailed description of the various elements which make up the system. The Regional Health Authorities, the Norwegian Knowledge Centre for Health Services, the Norwegian Medicines Agency and the Norwegian Directorate of Health are working closely together as regards the tasks linked to the establishment and implementation of the new system.
Systematic assessments of effect, safety, costs and consequences for patients and society can be ensured through the use of various types of HTAs based on internationally established principles for HTAs and research summaries [5, 6]. In HTAs, systematic summaries of available research on effect and safety are prepared (systematic overview) and a cost-effectiveness analysis is conducted in relation to a health service initiative, often together with an assessment of issues relating to ethics, law and the organisation of health services [7, 8]. HTA was formally established in Norway in 1997 with the creation of the Norwegian Centre for Health Technology Assessment [9], and has a long history in many other countries, including the USA, Canada, Australia, England, Scotland, Germany, France and Sweden [10-14].
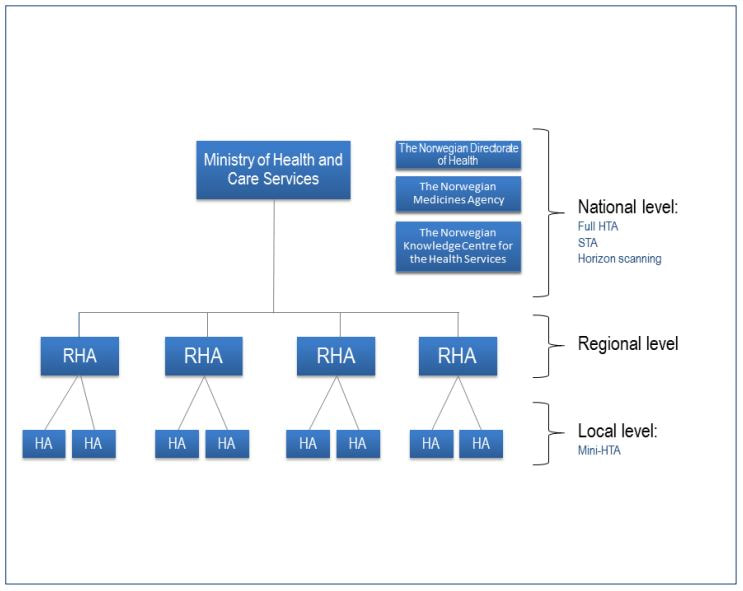
HTAs will provide a basis for qualified decisions regarding the introduction of new health technologies and will be carried out at various levels within the health service (Fig. 3). HTAs will act as pivotal tools for ensuring the safety and quality of patient care and sustainable development within the health services in the long term.
The development and implementation of health measures can be described as a life-cycle; from innovation and development to the introduction and establishment of new measures and possible withdrawal and replacement of measures that no longer meet adequate standards. During the course of such a life-cycle, there will be a need for various types of HTAs to support decision-makers in different phases and at various levels within the health service (see Figure 3). Many parties are involved in the various phases from the development of a new health technology to a decision concerning introduction or financing, development of guidelines and follow-up of service quality."
(Source: "The National system for the introduction of new health technologies within the specialist health service – For better and safer patient care" A working document from: The Regional Health Authorities, The Norwegian Medicines Agency, The Norwegian Knowledge Centre for Health Services, The Norwegian Directorate of Health. Version: 1.0 Date: 11 June 2013 / 23 January 2014, p. 13. PROJECT NUMBER: 9928, PROJECT MANAGER: Brynjar Fure; PARTICIPANT: FHI; ESTIMATED END DATE: Ongoing project; RESPONSIBLE DEPARTMENT: Kunnskapsoppsummering)
Figure 3 HTA’s at local, regional and national level. A mini-HTA is carried out at local level. If appropriate, issues may be channeled from the local hospital level through a regional level to a national process, in the form of a STA or full HTA. In particular cases the National Council for Priority Setting in Health Care may be consulted. (Source: ibid.)
- Background for the national system:
- Medical and healthcare technologies change quickly
- Variation or absence of routines for health technology assessment within the specialist health service
- Differences in decision-making processes related to new health technologies within the health care system
- "The purpose of the system (p. 14)
- Improve patient safety
- Ensure that patients gain equal access quickly to new methods that have proved to be effective and fulfill safety and cost-effective requirements
- Ensure that new methods that are ineffective and/or harmful are not introduced and that obsolete health technologies are disinvested
- Provide an appropriate decision-making platform for priority setting based on HTAs
- Ensure rational use of resources
- Establish a systematic and predictable process for the introduction of new methods"
- Systematic use of health technology assessments (HTA) to inform decision-making was the main ambition behind the establishment.
- "1.0 Summary
(...) "HTAs have been used for many years in numerous countries, including the USA, Canada, Australia, Sweden, England and Scotland, in addition to Norway. What is new in the Norwegian context is that HTAs are now being integrated into a holistic system with predictable and transparent processes for introducing new health technologies into the specialist health service. HTAs will be a tool for supporting appropriate prioritisation and decisions making in order to ensure that introduction of new technologies are proven as safe and effective (Figure 1). It will enable patients, health personnel and society in general to be certain that health technologies used in patient care are both safe and effective. The national system in its entirety will promote the rational use of resources within the health services." (p. 4)"
Norway's Initial HTA Agency 1997
The Norwegian Centre for Health Technology Assessment (Senter for medisinsk metodevurdering) (SMM), was established in 1997, as the initial HTA agency in Norway. The abstract of the Norwegian study, Mørland 2009, is included below.
- Abstract
Objectives: The aim of this study was to describe the Norwegian contribution to the broad picture of our international health technology assessment (HTA) history.
Methods: A general examination of some pre-HTA activities, as consensus conferences, closer reading of all background papers to the establishment of an HTA agency in Norway, the ongoing activities of this agency, its evaluation, and its merge into a broader knowledge center for the health services were performed. All information and statements are the authors’ own understandings, but also were reviewed by some of the people taken part in these processes.
Results: The Norwegian HTA activity was set up rather late compared to, for instance, Sweden, and our involvement in Nordic early warning activities. This may be related to Norwegian scientists being more involved in the Cochrane Collaboration. However, when established, the HTA agency was regarded as highly successful in involving clinical experts and responding to questions arising from the macro- as well as micro-levels of decision makers. International collaboration was essential in building the capacity to serve all levels. After 6 years, the agency was merged with some related health service knowledge activities, into a new center.
Conclusions: During the 10 years’ time, HTA has become a well-established activity in Norway. (Source: "The history of health technology assessment in Norway" Berit Mørland, The Norwegian Knowledge Centre for the Health Services. International Journal of Technology Assessment in Health Care, 25:Supplement 1 (2009), 148–155. doi:10.1017/S0266462309090576)
EUnetHTA
https://www.eunethta.eu/about-eunethta/mission-vision-and-values/
Update EUnetHTA JA3
https://ec.europa.eu/health/sites/health/files/technology_assessment/docs/ev_20170329_co07_en.pdf
INAHTA’S WORKING GROUP ON HANDLING ETHICAL ISSUES, Final report, INAHTA Ethics Working Group, June 2005
http://www.inahta.org/wp-content/uploads/2014/03/Final-report-Ethics-in-HTA-Nov-07.pdf
Using European Joint HTAs in local settings – to adopt or to adapt, that is… Katrine Frønsdal, BASc MSc PhD Norwegian Institute of Public Health (NIPH) Third WHO Global Forum on Medical Devices Centre International de Conférences, Genève (CICG) 10-12 May 2017
http://www.who.int/medical_devices/global_forum/3rd_gfmd/adoptoradapt.pdf
https://www.eunethta.eu/about-eunethta/mission-vision-and-values/
- Norway
Hdir
Norwegian Directorate of Health
NIPHNO (formerly NOKC)
The Norwegian Institute of Public Health
NOMA
Norwegian Medicines Agency
Update EUnetHTA JA3
https://ec.europa.eu/health/sites/health/files/technology_assessment/docs/ev_20170329_co07_en.pdf
INAHTA’S WORKING GROUP ON HANDLING ETHICAL ISSUES, Final report, INAHTA Ethics Working Group, June 2005
http://www.inahta.org/wp-content/uploads/2014/03/Final-report-Ethics-in-HTA-Nov-07.pdf
Using European Joint HTAs in local settings – to adopt or to adapt, that is… Katrine Frønsdal, BASc MSc PhD Norwegian Institute of Public Health (NIPH) Third WHO Global Forum on Medical Devices Centre International de Conférences, Genève (CICG) 10-12 May 2017
http://www.who.int/medical_devices/global_forum/3rd_gfmd/adoptoradapt.pdf
Ethics in Health Technology Assessments (HTA)
The purpose of the Norwegian study, Hofmann 2008, "is to present a practical method for addressing moral issues in HTA."
The "32 moral issues to be addressed" which were mentioned in the "Conclusion" section are presented in Table 2 (p. 39-40, included below).
The purpose of the Norwegian study, Hofmann 2008, "is to present a practical method for addressing moral issues in HTA."
- Abstract
"Background: Since the heyday of HTA it has been claimed that ethics is an integral part of HTA. However, few HTA reports have addressed moral issues, and there has been little agreement on methods for implementing ethics in HTA. Therefore, there is a need to develop a standard methodology for addressing ethical issues in HTA in the same manner as there is a methodology for systematic reviews and economic analysis.
Accordingly, the purpose of this report is to present a practical method for addressing moral issues in HTA. The method has to be applicable without special knowledge in ethics, and when applied, the method shall result in a broad basis for an open and transparent decision process. Accordingly, it shall only highlight the moral issues, and not lead to specific recommendations.
Method: On basis of analyses and assessments of existing methods in ethics in general and in HTA in particular, a practical procedure for addressing moral issues in HTA is to be developed and presented.
Results: A practical method for addressing moral issues in HTA is presented. The method consists of a procedure in six steps:
1. Identify and analyze the moral challenges with the health technology
2. Identify involved persons, groups, and stake holders
3. Identify relevant moral questions (from a list of questions) and justify the selection
4. Perform literature search in accordance with the identified moral questions
5. Analyze and discuss the moral questions identified (in step 3) on the basis of
a. The literature search
b. Hearings of involved parties or qualitative studies
6. Wrap up and summarize the process.
The core set of moral questions in step 3 belong to the following main groups:
1. General moral issues.
2. Moral issues related to stake-holders.
3. Moral issues related to the health technology.
4. Moral issues related to methodological choices in HTA.
5. Moral issues related to HTA itself.
Addressing the moral issues shall result in a broad basis for an open and transparent decision process.
Conclusion: A practical procedure has been developed for ethical analysis in HTA, based on an analysis of existing methods. The procedure is based on six steps and a selection of 32 moral issues to be addressed." (Source: Ethics in the assessment of health measures. Development of a methodology to visualize ethical challenges in assessing health measures. (Etikk i vurdering av helsetiltak. Utvikling av en metode for å synliggjøre etiske utfordringer ved vurdering av helsetiltak.) Nasjonalt kunnskapssenter for helsetjenesten (NOKC), Rapport fra Kunnskapssenteret nr 26 –2008, Ansvarlig John-Arne Røttingen, direktør; Forfatter: Bjørn Hofmann. ISBN 978-82-8121-223-7 ISSN 1890-1298)
The "32 moral issues to be addressed" which were mentioned in the "Conclusion" section are presented in Table 2 (p. 39-40, included below).
Table 1 Processual Processes Compared to Traditional Methods 18
(Tabell 1 Prosessuelle fremgangsmåter sammenliknet med tradisjonelle fremgangsmåter 18)
(Tabell 1 Prosessuelle fremgangsmåter sammenliknet med tradisjonelle fremgangsmåter 18)
|
(Source: ibid., p. 27)
"Table 2 (included below) provides a summary of morally relevant questions for use in method assessments."
Table 2: Morally relevant questions for use in assessing health measures
(Tabell 2 Moralsk relevante spørsmål for bruk i vurdering av helsetiltak)
(Tabell 2 Moralsk relevante spørsmål for bruk i vurdering av helsetiltak)
GENERAL MORAL QUESTIONS (GENERELLE MORALSKE SPØRSMÅL) 1 What is the problem that the health measure should solve? Who defines the problem and solution options? (Also describe the options.) (Hva er problemet som helsetiltaket skal løse? Hvem definerer problemet og løsningsalternativer? (Beskriv også alternativene.)) |
2 What are the morally relevant consequences of using the health measures? (Include risk, uncertainty and areas of ignorance that may be associated with technology.) (Hva er de moralsk relevante konsekvensene av å ta i bruk helsetiltaket? (Inkluder risiko, usikkerhet og områder for uvitenhet som kan knyttes til teknologien.)) |
3 Challenges the introduction or use of healthcare patients' autonomy, their integrity, dignity or their (human) rights? (Utfordrer innføring eller bruk av helsetiltaket pasientenes autonomi, deres integritet, verdighet eller deres (menneske)rettigheter?) |
4 Does the moral obligation follow the introduction and use of the health measure? (Følger det moralske forpliktelser med å innføre og bruke helsetiltaket?) |
5 Challenges health measures social values, schemes or institutions? (Utfordrer helsetiltaket sosiale verdier, ordninger eller institusjoner?) |
6 Does extensive use of health measures change our perception of specific persons or groups (for example, persons with specific diagnoses)? (Forandrer utstrakt bruk av helsetiltaket vår oppfatning av bestemte personer eller grupper (for eksempel personer med bestemte diagnoser)?) |
7 Does the health measure be offensive against religious, social or cultural beliefs? (Virker helsetiltaket støtende i forhold til religiøse, sosiale eller kulturelle overbevisninger?) |
8 Does the health measure challenge relevant laws? (Utfordrer helsetiltaket relevante lover?) |
9 How does the current health measure relate to general criticisms of modernization medicine (for example, medication)? (Hvordan forholder det aktuelle helsetiltaket seg til generelle kritikker av moderne medisin (for eksempel medikalisering)?) |
10 Are there any related measures that have proven to be morally controversial? (Finnes det beslektede tiltak som har vist seg å være moralsk kontroversielle?) |
11 Does the health measure in any way challenge or change the relationship between a fellow and a patient? (Utfordrer eller forandrer helsetiltaket på noen måte forholdet mellom pelsepersonell og pasient?) |
12 How does the implementation and use of health services affect the distribution of healthcare? (Hvordan påvirker implementering og bruk av helsetiltaket fordelingen av helsetjenester?) |
13 How does the health measure help to strengthen or impair professional self-determination? (Hvordan bidrar helsetiltaket til å styrke eller svekke profesjonell selvbestemmelse?) |
14 Can health measures hurt the patient? (Kan helsetiltaket skade pasienten?) |
QUESTIONS WORKING TO ANY INTERESTED PARTIES (SPØRSMÅL KNYTTET TIL ULIKE INTERESSENTER) 15 Which patient groups benefit from the health measures? (Hvilke pasientgrupper har nytte av helsetiltaket?) |
16 Are third parties involved and, if so, their interests? (Er tredjeparter involvert, og hva er i så fall deres interesser?) |
17 What are the interests of those who use the health measures? (Hvilke interesser har de som bruker helsetiltaket?) |
18 What are the interests of the manufacturers of the health measures? (Industry, University) (Hvilke interesser har produsentene av helsetiltaket? (Industri, universitet)) |
QUESTIONS WORKING TO THE HEALTH ADVANTAGE (SPØRSMÅL KNYTTET TIL HELSETILTAKET) 19 Are moral challenges related to parts of the health measures relevant to the health measures as a whole? (Er moralske utfordringer knyttet til deler av helsetiltaket relevante for helsetiltaket som helhet?) |
20 What is characteristic of the health measures that are considered? (Hva er karakteristisk for helsetiltaket som vurderes?) |
21 Does the symbolic value of the health measure have any moral relevance? (Har helsetiltakets symbolske verdi noen moralsk relevans?) |
QUESTIONS ON MORAL ASPECTS BY METHODOLOGICAL OPTIONS (SPØRSMÅL OM MORALSKE ASPEKTER VED METODOLOGISKE VALG) 22 Is the morally relevant question related to the choice of endpoints in the methodological assessment? (Er det moralsk relevante spørsmål knyttet til valg av endepunkter i den metodiske vurderingen?) |
23 Are there any morally relevant questions related to the inclusion of studies in the method assessment? (Er det moralsk relevante spørsmål knyttet til inklusjonen av studier i metodevurderingen?) |
24 Are the users in the studies representative of those who want to use health measures in clinical practice? (Er brukerne i studiene representative for dem som vil bruke helsetiltaket i klinisk praksis?) |
25 Are there morally relevant aspects related to how knowledge is generalized? (Er det moralsk relevante aspekter knyttet til hvordan kunnskapen generaliseres? (25 Er det moralsk relevante aspekter knyttet til hvordan kunnskapen generaliseres?) |
26 Are there ethical research challenges related to the method assessment? (Finnes det forskningsetiske utfordringer knyttet til metodevurderingen?) |
QUESTIONS WORKED TO IMPLEMENT A METHOD ASSESSMENT (HTA) (SPØRSMÅL KNYTTET TIL Å GJENNOMFØRE EN METODEVURDERING (HTA)) 27 What is the reason for this health measure being considered (possibly in relation to other measures)? (Hva er grunnen til at dette helsetiltaket vurderes (eventuelt i forhold til andre tiltak)?) |
28 Is the method assessment suitable to elucidate all relevant aspects of the health measure? (Er metodevurderingen egnet til å belyse alle relevante aspekter ved helsetiltaket?) |
29 What interests do those involved in the assessment have? (Hvilke interesser har de som deltar i vurderingen?) |
30 At what stage in the development of health measures, is the method assessment carried out? (På hvilket stadium i utviklingen av helsetiltaket blir metodevurderingen gjennomført?) |
31 Are there any related measures that are not considered? (Finnes det beslektede tiltak som ikke er vurdert?) |
32 What are the moral consequences of the method assessment? (Hva er metodevurderingens moralske konsekvenser?) |
Attachment: Map of Value Analysis (Vedlegg: Kort om verdianalyse)
The approach is based on a value analysis (axiology) that understands health technology as part of human activity and that it is related to values in different ways. 25
Health technology can have value through its:
(Verdier gjør seg gjeldende på mange måter med hensyn til innføring og bruk av helseteknologi:)
Interestingly, the excerpt below discusses the ethical and moral issues of the fetal ultrasound screening program.
The approach is based on a value analysis (axiology) that understands health technology as part of human activity and that it is related to values in different ways. 25
Health technology can have value through its:
- Function: For example, the function of a diagnostic ultrasound device is to capture images of structures within the body using ultrasonic reflections. (funksjon: Eksempelvis er funksjonen til et diagnostisk ultralydapparat å ta bilder av strukturer inne i kroppen ved hjelp av ultralydrefleksjoner.)
- Purpose: The purpose of an ultrasound device may be to gain knowledge about structures within the body. (hensikt: Hensikten med et ultralydapparat kan være å få kunnskap om strukturer inne i kroppen.)
- Intention: A technology for a given purpose can be used for different purposes: For example, the value of acquiring knowledge about structures within the body using diagnostic ultrasound apparatus is to treat the patient. (intensjon: En teknologi med en gitt hensikt kan brukes til ulike formål: Verdien av å få kunnskap om strukturer inne i kroppen ved hjelp diagnostisk ultralydapparat er eksempelvis at man kan behandle pasienten.)
- Higher Order Intention 26: Diagnostic Ultrasound, for example, has a professional status which for too many radiologists is lower than other imaging techniques, such as CT, MR and PET. These types of values will also apply when the technology is assessed, implemented and used. (høyere ordens intensjon 26: Diagnostisk ultralyd har eksempelvis en faglig status som for mange radiologer er lavere enn andre bildedannende teknikker, som CT, MR og PET. Denne typen verdier vil også gjøre seg gjeldende når teknologien vurderes, implementeres og anvendes.)
(Verdier gjør seg gjeldende på mange måter med hensyn til innføring og bruk av helseteknologi:)
- General moral aspects: Consequences, self-determination, integrity, dignity, human rights. For example, fetal diagnostic ultrasound can increase women's self-determination with regard to having children. At the same time it can lead to changed perceptions of people who have conditions that many perceive as abortion. It can also challenge perceptions of dignity and actualization the question of what rights fetuses have. (Generelle moralske aspekter: Konsekvenser, selvbestemmelse, integritet, verdighet, menneskerettigheter. Eksempelvis kan fosterdiagnostisk ultralyd øke kvinners selvbestemmelse med hensyn på å få barn. Samtidig kan det føre til endrede oppfatninger av personer som har tilstander som av mange oppfattes som abortgrunn. Det kan også utfordre oppfatninger av verdighet og aktualisere spørsmåltet om hvilke rettigheter fostre har.)
- Aspects related to interests: Professional, industry, patient (organization) is, users, health technologists, authorities. 27 Manufacturers and professionals have interests of mass surveys of ultrasound fosters. (Aspekter relatert til interesser: Profesjonelle, industri, pasient(organisasjon)er, brukere, helseteknologivurderere, myndigheter. 27 Produsenter og fagfolk har interesser av masseundersøkelser av fostre med ultralyd.)
- Aspects related to methodological choices: Endpoints, evidence level. How should you consider the yield of, for example, ultrasound in fetal diagnostics: In number detected cases of Down's syndrome, number of abortions of fetal non-Down syndrome (false positive), number of abortions, number of people with Down syndrome in society? (Aspekter relatert til metodologiske valg: Endepunkter, evidensnivå. Hvordan skal man vurdere utbyttet av eksempelvis ultralyd i fosterdiagnostikk: I antall oppdagede tilfeller av Downs syndrom, antall aborter av fostre uten Downs syndrom (falsk positive), antall aborter, antall personer med Downs syndrom i samfunnet?)
- Aspects related to methodology and health policy: Selection of health technology to be assessed. 28 What is the reason why ultrasound is subject to method assessment? (Aspekter relatert til metodevurdering og helsepolitikk: Valg av helseteknologi som skal vurderes. 28 Hva er grunnen til at ultralyd er gjenstand for metodevurdering?)
Interestingly, the excerpt below discusses the ethical and moral issues of the fetal ultrasound screening program.
- Does it follow moral obligations to introduce and use the health measure?
(Følger det moralske forpliktelser med å innføre og bruke helsetiltaket?)
The question relates to the imperatives of possibilities (91). When anything becomes possible, it often leads to pressure that it will be implemented. This can be morally challenging, for example, if a diagnostic method is introduced without adequate therapeutic measures. Similarly, women who for various reasons do not want to participate in the ultrasound screening program for fetuses may experience attitudes like "it's your own fault" if children are born with developmental impairment (69). (Spørsmålet relaterer seg til mulighetenes imperativ (91). Når noe blir mulig, fører det ofte til press om at det skal gjennomføres. Dette kan være moralsk utfordrende, for eksempel om en diagnostisk metode innføres uten at det finnes adekvate terapeutiske tiltak. Tilsvarende kan man oppleve at kvinner som av ulike grunner ikke ønsker å delta i ultralydscreeningprogrammet for fostre, kan oppleve holdninger som at ”det er din egen feil” dersom barn blir født med utviklingshemming (69).)
Health measures may cause other obligations (and side effects) than planned, but as morally challenging. Attempting to take this into account when assessing health measures seems to be important. This question has its headings mainly in procedural methods. (Helsetiltak kan medføre andre forpliktelser (og bieffekter) enn planlagt, men som er moralsk utfordrende. Å forsøke å ta hensyn til dette i forbindelse med vurderingen av helsetiltak, synes å være viktig. Dette spørsmålet har sitt utspring hovedsakelig i prosessuelle fremgangsmåter.) (Source: Ethics in the assessment of health measures. Development of a methodology to visualize ethical challenges in assessing health measures. (Etikk i vurdering av helsetiltak. Utvikling av en metode for å synliggjøre etiske utfordringer ved vurdering av helsetiltak.) Nasjonalt kunnskapssenter for helsetjenesten.Rapport fra Kunnskapssenteret nr 26 –2008, p. 30-31. Ansvarlig John-Arne Røttingen, direktør; Forfatter: Bjørn Hofmann. ISBN 978-82-8121-223-7 ISSN 1890-1298)
NOKC & Evidence-informed Health Policy (EIHP) Using Research
Interestingly, a 2008 study authored by members of the Norwegian Knowledge Centre for the Health Services (NOKC) explicitly identified: 1) NOKC's purpose with respect to "Evidence-informed Health Policy Using Research" and 2) NOKC's claimed scientific and professional independence from Directorate of Health, i.e., before "the fix.". [Note: In January 2016, NOKC separated from the Norwegian Directorate of Health and joined the Norwegian Institute of Public Health (NIPH) and is now NIPH Division of Health Services.]
The Norwegian Knowledge Centre for the Health Services (NOKC) published a 283-page book titled: "SUPPORT Tools for evidence-informed health Policymaking (STP)" which stated: "Knowing how to find and use research evidence can help policymakers and those who support them to do their jobs better and more efficiently. Each chapter presents a proposed tool that can be used by those involved in finding and using research evidence to support evidence-informed health policymaking. The book addresses four broad areas:
Interestingly, a 2008 study authored by members of the Norwegian Knowledge Centre for the Health Services (NOKC) explicitly identified: 1) NOKC's purpose with respect to "Evidence-informed Health Policy Using Research" and 2) NOKC's claimed scientific and professional independence from Directorate of Health, i.e., before "the fix.". [Note: In January 2016, NOKC separated from the Norwegian Directorate of Health and joined the Norwegian Institute of Public Health (NIPH) and is now NIPH Division of Health Services.]
- "Norwegian Knowledge Centre for the Health Services summarizes and disseminates evidence concerning the effect of treatments, methods, and interventions in health services, in addition to monitoring health service quality. Our goal is to support good decision making in order to provide patients in Norway with the best possible care. The Centre is organized under The Directorate for Health and Social Affairs, but is scientifically and professionally independent. The Centre has no authority to develop health policy or responsibility to implement policies." (Source: "Evidence-Informed Health Policy: Using Research to Make Health Systems Healthier" Ray Moynihan, Andrew David Oxman, John N. Lavis, and Elizabeth Paulsen Rapport Nr 1-2008, Project number 265. Oslo: Nasjonalt kunnskapssenter for helsetjenesten, January 2008. ISBN 978-82-8121-187-2, ISSN 1890-1298)
- "The study’s seven main implications for those establishing or administering organisations to produce clinical practice guidelines or health technology assessments or organisations to support the use of research evidence in developing health policy include:
1. Collaborate with other organisations
2. Establish strong links with policymakers and involve stakeholders in the work
3. Be independent and manage conflicts of interest among those involved in the work
4. Build capacity among those working in the organisation
5. Use good methods and be transparent in the work
6. Start small, have a clear audience and scope, and address important questions
7. Be attentive to implementation considerations even if implementation is not a remit"
(Source: ibid.) - "The study's four main implications for the World Health Organisation and other international organisations include:
1. Support collaborations among organisations
2. Support local adaptation efforts
3. Mobilize support
4. Create knowledge-related global public goods, including methods and evidence syntheses"
(Source: ibid.)
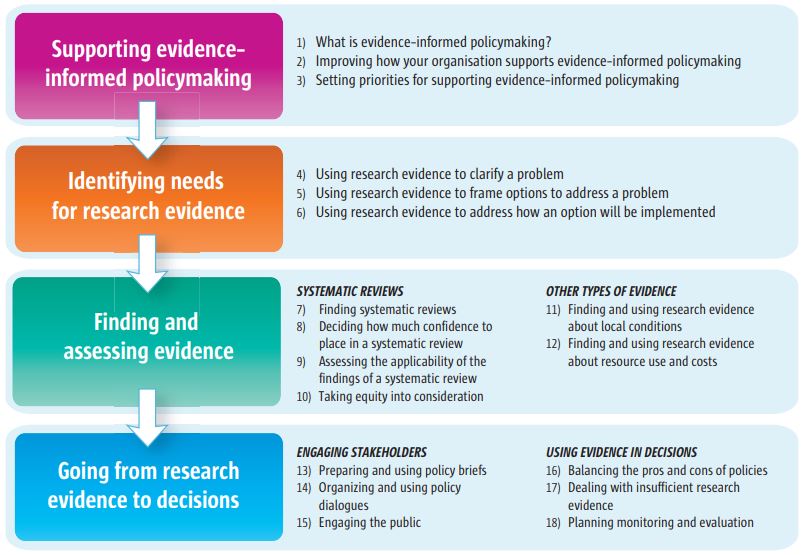
The Norwegian Knowledge Centre for the Health Services (NOKC) published a 283-page book titled: "SUPPORT Tools for evidence-informed health Policymaking (STP)" which stated: "Knowing how to find and use research evidence can help policymakers and those who support them to do their jobs better and more efficiently. Each chapter presents a proposed tool that can be used by those involved in finding and using research evidence to support evidence-informed health policymaking. The book addresses four broad areas:
- Supporting evidence-informed policymaking,
- Identifying needs for research evidence in relation to three steps in policymaking processes, namely problem clarification, options framing, and implementation planning,
- Finding and assessing both systematic reviews and other types of evidence to inform these steps, and
- Going from research evidence to decisions."
(Source: "SUPPORT Tools for evidence-informed health Policymaking (STP)" Report from Norwegian Knowledge Centre for the Health Services (Nasjonalt kunnskapssenter for helsetjenesten) No 4–2010)
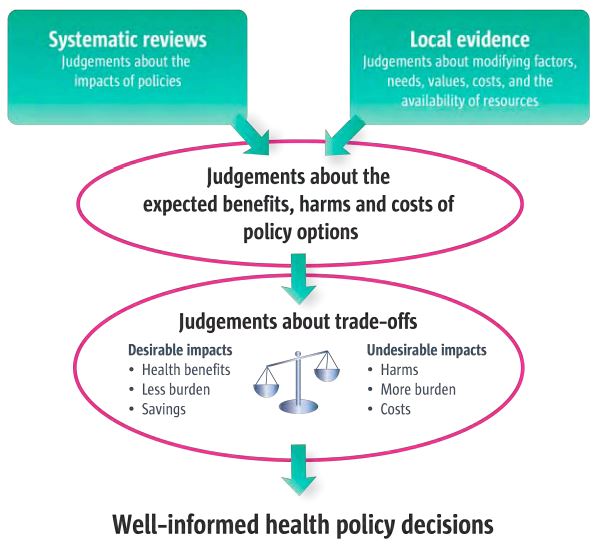
Figure 1.1 An example of the role of evidence in health policymaking (p. 21)
Figure 1.2 How evidence-informed health policymaking addresses common policymaking problems (p. 24)
The following statement is incluided on page xxx of the Knowledge Center's Håndbok for Nasjonalt kunnskapssenter for helsetjenesten desember 2015:"This is how we summarize research" ("Slik oppsummerer vi forskning")
Nasjonalt kunnskapssenter for helsetjenesten fremskaffer og formidler kunnskap om effekt av metoder, virkemidler og tiltak og om kvalitet innen alle deler av helsetjenesten. Målet er å bidra til gode beslutninger slik at brukerne får best mulig helsetjenester. Inntil 31.12.2015 er senteret formelt et forvaltningsorgan under Helsedirektoratet, uten myndighetsfunksjoner. Kunnskapssenteret kan ikke instrueres i faglige spørsmål. Fra 2016 er Kunnskapssenteret innlemmet i Folkehelseinstituttet.
ENGLISH
NOKC Independence and Legitimacy
The Knowledge Center's independence and legitimacy was identified as an issue in the summary section of the 2007 report: "Evaluation of the National Knowledge Center for the Health Services - Final Report" ("Evaluering av Nasjonalt kunnskapssenter for helsetjenesten - Sluttrapport").
- "The National Knowledge Center for Health Services provides and disseminates knowledge of the effects of methods, measures and measures of quality in all parts of the health service. The goal is to make good decisions so that users get the best possible healthcare. Until 31.12.2015, the Center is formally a governing body under the Directorate of Health, without governmental functions. The Knowledge Center can not be instructed in academic matters. From 2016, the Knowledge Center is incorporated into the National Institute of Public Health."
Nasjonalt kunnskapssenter for helsetjenesten fremskaffer og formidler kunnskap om effekt av metoder, virkemidler og tiltak og om kvalitet innen alle deler av helsetjenesten. Målet er å bidra til gode beslutninger slik at brukerne får best mulig helsetjenester. Inntil 31.12.2015 er senteret formelt et forvaltningsorgan under Helsedirektoratet, uten myndighetsfunksjoner. Kunnskapssenteret kan ikke instrueres i faglige spørsmål. Fra 2016 er Kunnskapssenteret innlemmet i Folkehelseinstituttet.
ENGLISH
NOKC Independence and Legitimacy
The Knowledge Center's independence and legitimacy was identified as an issue in the summary section of the 2007 report: "Evaluation of the National Knowledge Center for the Health Services - Final Report" ("Evaluering av Nasjonalt kunnskapssenter for helsetjenesten - Sluttrapport").
- "The Centre’s Independence and Legitimacy
"The Centre’s independence within its field of activity and its scientific legitimacy are essential components for its success. The evaluation team feels that the Centre’s legitimacy has grown over time and that the Centre appears to be well accepted. In many associated fields and among relevant actors, scepticism has been expressed regarding the newly established secretarial function for the National Council for Quality Improvement and Prioritizing in the Health Services. Independence and legitimacy require a continual balancing act in relation to political guidelines." (p. 6)
"Kunnskapssenterets uavhengighet og legitimitet
Kunnskapssenterets faglige uavhengighet og vitenskapelige legitimitet er helt avgjørende. Legitimiteten synes for evalueringsgruppen å ha økt med tiden og Kunnskapssenteret synes å være godt akseptert. Det er i mange fagmiljøer og blant relevante aktører uttrykt skepsis til den nyetablerte sekretariatsfunksjonen for Nasjonalt råd for kvalitet og prioritering. Uavhengighet og legitimitet krever en kontinuerlig balansegang i forhold til politiske føringer." (p. 6)
(Source: "Evaluering av Nasjonalt kunnskapssenter for helsetjenesten - Sluttrapport" Carried out in 2007 under the leadership of Johan Calltorp on behalf of the Directorate for Health and Social Affairs. Utgitt: 15.oktober 2007, Bestillingsnr.: IS – 1503, Avgitt til: Sosial- og helsedirektoratet.) - "There is a great risk that trust that is built up as an impartial organization for knowledge summary without any ambition other than presenting facts can be quickly broken down if the organization also conducts political or semi-political assessments with recommendations of a common nature."
"Det er en stor risiko for at tilliten som er bygget opp som en upartisk organisasjon for kunnskapsoppsummering uten andre ambisjoner enn å fremlegge fakta, raskt kan brytes ned dersom organisasjonen også foretar politiske eller semipolitiske vurderinger med anbefalinger av normerende art." (Source: ibid., p. 19) - "4.6 Knowledge Center's independence, trust and visibility
"The Knowledge Center's professional independence is attached to the articles of association: "The Knowledge Center is a professional independent administrative body without governmental functions. The Knowledge Center can not be instructed in academic questions. Knowledge Center determines their own form of work, communicates freely and publishes their work." The knowledge center is, however, subject to SHdir in level three under Ministry of Health and Care Services. SHdir is also an important principal and chief finance officer."
"4.6 Kunnskapssenterets uavhengighet, tillit og synlighet.
Kunnskapssenterets faglige uavhengighet er vedtektsfestet: ”Kunnskapssenteret er et faglig uavhengig ordinært forvaltningsorgan uten myndighetsfunksjoner. Kunnskapssenteret kan ikke bli instruert i faglige spørsmål. Kunnskapssenteret bestemmer egen arbeidsform, formidler fritt og offentliggjør sitt arbeid.” Kunnskapssenteret er imidlertid underlagt SHdir i nivå tre under Helse- og omsorgsdepartementet. SHdir er samtidig en viktig oppdragsgiver og hovedfinansiør." (Source: ibid., p. 49) - "Special representatives of academic and health services are still skeptical of the administrative ties they perceive between the Knowledge Center and SHdir which is agency governor, ordering, financier and decision maker. This gives background for assessing whether the professional independence is real. Regional Health Authorities' professionals have generally expressed questioned and criticized the connection with SHdir via agency but see that it has also had positive sides. It was also pointed out that Haste assignments can threaten the Knowledge Center's professional independence because they affect Priorities in the Knowledge Center."
"Særlig representanter for fagmiljøer og helsetjenesten er fortsatt skeptiske til de administrative bindingene de mener å oppfatte mellom Kunnskapssenteret og SHdir som er både etatsstyrer, bestiller, finansiør og beslutningstaker. Dette gir bakgrunn for vurderinger av hvorvidt den faglige uavhengigheten er reell. RHFenes fagdirektører har generelt uttalt seg spørrende og kritiske til tilknytningen til SHdir via etatsstyring, men ser at den også har hatt positive sider. Det ble for øvrig påpekt at hasteoppdrag kan true Kunnskapssenterets faglige uavhengighet, fordi de påvirker prioriteringene i Kunnskapssenteret." (Source: ibid., p. 50) - "Evaluation Group recommendations
Systematic and open process for prioritizing key issues
The Evaluation Group recommends that the Knowledge Center develop a systematic process for identifying key issues within all important disciplines and levels of health services. The purpose is to ensure strategically correct theme choices and to meet the needs of the target groups. The process should be open: it should be clear who participates, what criteria are the basis for the decisions and the content of these. All relevant clients and user groups should be involved in the process."
"Evalueringsgruppens anbefalinger
Systematisk og åpen prosess for prioritering av sentrale problemstillinger
Evalueringsgruppens anbefalinger
Evalueringsgruppen anbefaler at Kunnskapssenteret utvikler en systematisk prosess for å identifisere sentrale problemstillinger innen alle viktige fagområder og nivåer av helsetjenesten. Hensikten er å sikre strategisk riktige temavalg, og at man treffer målgruppenes behov. Prosessen bør være åpen: det bør fremgå tydelig hvem som deltar, hvilke kriterier som legges til grunn for beslutningene, og innholdet i disse. Alle relevante oppdragsgivere og brukergrupper bør bli involvert i prosessen." (Source: ibid., p. 35) - "Clear balance between leadership and own priorities
A good prioritization process assumes that the guidelines of the ministry and the governors are kept at an overall level. The evaluation group recommends that the agency and the Knowledge Center clarify the balance between professional independence in the prioritization process and the consideration of policy guidance through the agency management."
"Tydeliggjort balansegang mellom føringer og egne prioriteringer
En god prioriteringsprosess forutsetter at føringene fra departementet og etatsstyrer holdes på et overordnet nivå. Evalueringsgruppen anbefaler at etatsstyrer og Kunnskapssenteret tydeliggjør balansegangen mellom faglig uavhengighet i prioriteringsprosessen og hensynet til politiske føringer gjennom etatsstyringen." (Source: ibid., p.60)
NOKC: Was Not"Scientifically and Professionally Independent" of Directorate of Health
Included below is the Wikipedia entry for Cecilie Daae, MD who left Directorate of Health to be the Director, Norwegian Directorate for Civil Protection (Direktoratet for samfunnssikkerhet og beredskap) (DSB) on 01.09.2015, reporting to the Ministry of Justice and Public Security. The Wikipedia entry for Cecilie Daae describes her employment with Directorate of Health which included her work with NOKC regarding "National systems for assessing new methods and medicines in the specialist health service." Cedilie Daae's work with NOKC while an employee of Directorate of Health would appears to have been work which was both professional and scientific.
Cecilie Daae is credited as a member of the Ordering Forum RHF as follows: "Medlem: Helsedirektoratet v/ divisjonsdirektør Cecilie Daae" (p. 10) in the "Annual Report 2013: National system for introducing new methods in the specialist health service" ("Årsrapport 2013: Nasjonalt system for innføring av nye metoder i spesialisthelsetjenesten"). And, Cecilie Daae's professional and scientific work for NOKC contributed to the creation and 2015 publication of Ministry of Health and Care Services' New Methods System (Nye Metoder System) which, at the time, was known by its original, formal & much-too-long title (below):
Included below is the Wikipedia entry for Cecilie Daae, MD who left Directorate of Health to be the Director, Norwegian Directorate for Civil Protection (Direktoratet for samfunnssikkerhet og beredskap) (DSB) on 01.09.2015, reporting to the Ministry of Justice and Public Security. The Wikipedia entry for Cecilie Daae describes her employment with Directorate of Health which included her work with NOKC regarding "National systems for assessing new methods and medicines in the specialist health service." Cedilie Daae's work with NOKC while an employee of Directorate of Health would appears to have been work which was both professional and scientific.
- "Cecilie Daae (born 1962 ) is director of the Directorate for Social Security and Preparedness . [1]
("Cecilie Daae (født 1962) er direktør i Direktoratet for samfunnssikkerhet og beredskap. [1]")
She has a medical diploma from the University of Oslo [1] 1990, was approved as a specialist in general medicine in 2001 and re-certified in 2006. Daae has previously served as division director in the specialist division and department director in the department of hospital services in the Norwegian Directorate of Health before she was employed temporarily as one of two Assistant Directors of the Directorate of Health from April 30, 2014. [1] [2] On September 1, 2015, Daae joined the position as Director of DSB, thereby becoming the second director in the line for the agency established in 2005.
("Hun har medisinsk embetseksamen fra Universitetet i Oslo[1] 1990, ble godkjent som spesialist i allmennmedisin i 2001 og resertifisert i 2006. Daae har tidligere vært divisjonsdirektør i spesialistdivisjonen og avdelingsdirektør i avdeling sykehustjenester i Helsedirektoratet, før hun ble ansatt midlertidig som en av to assisterende direktører i Helsedirektoratet fra 30. april 2014.[1][2] Den 1. september 2015 tiltrådte Daae stillingen som direktør for DSB, og ble med dette den andre direktøren i rekken for etaten som ble opprettet i 2005.")
"For the Directorate of Health, Daae also worked with the management of the National Knowledge Center for the Health Services and was responsible for the Directorate's work with, among other things, emergency networks and national systems for assessing new methods and medicines in the specialist health service."
("For Helsedirektoratet arbeidet Daae også med etatsstyring av Nasjonalt kunnskapssenter for helsetjenesten og var ansvarlig for direktoratets arbeid med blant annet nødnett og nasjonalt system for vurdering av nye metoder og legemidler i spesialisthelsetjenesten.")
(Source: Wikipedia: https://no.wikipedia.org/wiki/Cecilie_Daae)
Cecilie Daae is credited as a member of the Ordering Forum RHF as follows: "Medlem: Helsedirektoratet v/ divisjonsdirektør Cecilie Daae" (p. 10) in the "Annual Report 2013: National system for introducing new methods in the specialist health service" ("Årsrapport 2013: Nasjonalt system for innføring av nye metoder i spesialisthelsetjenesten"). And, Cecilie Daae's professional and scientific work for NOKC contributed to the creation and 2015 publication of Ministry of Health and Care Services' New Methods System (Nye Metoder System) which, at the time, was known by its original, formal & much-too-long title (below):
- English version: "The National system for the introduction of new health technologies within the specialist health service – For better and safer patient care" (See: New Methods System)
- Norwegian version: "Nasjonalt system for innføring av nye metoder i spesialisthelsetjenesten – for bedre og tryggere pasientbehandling" (See: New Methods System)
- "Daae has previously served as division director in the specialist division and department director in the department of hospital services in the Norwegian Directorate of Health..." (Source: Wikipedia: https://no.wikipedia.org/wiki/Cecilie_Daae)
Evidence-informed Health Policymaking (EIHP)
[Note: Norwegian institutions and authors identified in red text below.]
[Note: Norwegian institutions and authors identified in red text below.]
- "SUPPORT Tools for evidence-informed health Policymaking (STP)" Edited by Andy Oxman and Stephen Hanney
- "This series of articles was prepared as part of the SUPPORT project, which was supported by the European Commission’s 6th Framework INCO programme, contract 031939. The Norwegian Agency for Development Cooperation (Norad), the Alliance for Health Policy and Systems Research, and the Milbank Memorial Fund funded a peer review meeting where an earlier version of the series was discussed. The Canada Research Chairs Program provides salary support to John Lavis, who has authored several articles in this series, in his role as Chair Canada Research Chair in Knowledge Transfer and Exchange. None of the funders had a role in drafting, revising or approving the content of this series."
- Authors: John N Lavis *1, Andrew D Oxman 2, Simon Lewin 3 and Atle Fretheim 4
- Address: 1 Centre for Health Economics and Policy Analysis, Department of Clinical Epidemiology and Biostatistics, and Department of Political Science, McMaster University, 1200 Main St. West, HSC-2D3, Hamilton, ON, Canada, L8N 3Z5, 2 Norwegian Knowledge Centre for the Health Services, P.O. Box 7004, St. Olavs plass, N-0130 Oslo, Norway, 3 Norwegian Knowledge Centre for the Health Services, P.O. Box 7004, St. Olavs plass, N-0130 Oslo, Norway; Health Systems Research Unit, Medical Research Council of South Africa and 4 Norwegian Knowledge Centre for the Health Services, P.O. Box 7004, St. Olavs plass, N-0130 Oslo, Norway; Section for International Health, Institute of General Practice and Community Medicine, Faculty of Medicine, University of Oslo, Norway
- What is evidence-informed policymaking? (Full Text or PDF)
- Improving how your organisation supports the use of research evidence to inform policymaking (Full Text or PDF)
- Setting priorities for supporting evidence-informed policymaking (Full Text or PDF)
- Using research evidence to clarify a problem (Full Text or PDF)
- Using research evidence to frame options to address a problem (Full Text or PDF)
- Using research evidence to address how an option will be implemented (Full Text or PDF)
- Finding systematic reviews (Full Text or PDF)
- Deciding how much confidence to place in a systematic review (Full Text or PDF)
- Assessing the applicability of the findings of a systematic review (Full Text or PDF)
- Taking equity into consideration when assessing the findings of a systematic review (Full Text or PDF)
- Finding and using evidence about local conditions (Full Text or PDF)
- Finding and using research evidence about resource use and costs (Full Text or PDF)
- Preparing and using policy briefs to support evidence-informed policymaking (Full Text or PDF)
- Organising and using policy dialogues to support evidence-informed policymaking (Full Text or PDF)
- Engaging the public in evidence-informed policymaking (Full Text or PDF)
- Using research evidence in balancing the pros and cons of policies (Full Text or PDF)
- Dealing with insufficient research evidence (Full Text or PDF)
- Planning monitoring and evaluation of policies (Full Text or PDF)
(Source: "SUPPORT Tools for evidence-informed health Policymaking (STP)" Health Research Policy and Systems, Volume 7 Supplement 1. https://health-policy-systems.biomedcentral.com/articles/supplements/volume-7-supplement-1; All-in-one PDF document published by NOKC: https://www.fhi.no/globalassets/kss/filer/filer/publikasjoner/rapporter/20102/stp-rapport-4-2010_web.pdf)
Figure 1: Overview of the series
(Source: "SUPPORT Tools for evidence-informed health Policymaking (STP)" Report from Norwegian Knowledge Centre for the Health Services No 4–2010, Chapter 1, p. 25. ISBN print 978-82-8121-313-5, ISBN digital 978-82-8121-334-0, ISSN 1890-1298; Or see: "Introduction: SUPPORT Tools for evidence-informed health Policymaking (STP)" John N Lavis, Andrew D Oxman, Simon Lewin and Atle Fretheim. Published: 16 December 2009. Health Research Policy and Systems 2009, 7(Suppl 1):I1 doi:10.1186/1478-4505-7-S1-I1. http://www.health-policy-systems.com/content/7/S1/I1 or PDF)
"What evidence-informed policymaking is not
Like any other tool, those that are used to support the use of evidence to inform policymaking can be misused. Undesirable impacts arising from the inappropriate use of evidence can include inefficient bureaucratic processes, the inappropriate inhibition or delay of promising programmes, the misleading framing of problems, the manipulation of public opinion, and the distortion of the research agenda. Ways in which evidence can be misused include using evidence selectively, stifling the appropriate use of evidence, and creating of a spurious impression of uncertainty. The best way to detect and prevent the inappropriate use of evidence is to use processes that are systematic and transparent, as we will describe in subsequent articles in this series." (Source: "SUPPORT Tools for evidence-informed health Policymaking (STP)" Report from Norwegian Knowledge Centre for the Health Services No 4–2010, Chapter 1, p. 25. ISBN print 978-82-8121-313-5, ISBN digital 978-82-8121-334-0, ISSN 1890-1298)
"Conclusion
The SUPPORT tools in this series have been designed to help policymakers and those who support them to do one aspect of their job better or more efficiently – namely to find and use research evidence to support health policymaking. The tools are also relevant to health system stakeholders, such as non-governmental organisations and civil society groups. Different readers will use the tools in different ways. Policymakers may skim the articles to get ideas on how they should be adjusting the expectations they set for their staff. Those who support policymakers may choose to read a particular article to help them with undertaking a new activity, and then use the article later as a reference guide or as a way of refining their skills. We hope that policymakers and those who support them will help us to develop and improve what is presented here." (Source: ibid., p. 25)
Evidence-informed Health Policymaking (EIHP) Was Ignored
It is apparent NOKC's efforts and experience developing"SUPPORT Tools for evidence-informed health Policymaking (STP)" were not applied to Directorate of Health's knowledge-obviated, medically & ethically flawed 2014 Recommendation with their exclusive implementation of NCFM eSnurra Group's "method" (i.e., the appropriated, plagiarized, misused Hutchon Method of PDEE) within a government-mandated protocol of evidence-obviated medicine; a protocol proven to cause increased medical risks, critical medical mistakes and grievous medical harms with respect to obstetric medicine, fetal medicine and obstetric clinical care.
"What evidence-informed policymaking is not
Like any other tool, those that are used to support the use of evidence to inform policymaking can be misused. Undesirable impacts arising from the inappropriate use of evidence can include inefficient bureaucratic processes, the inappropriate inhibition or delay of promising programmes, the misleading framing of problems, the manipulation of public opinion, and the distortion of the research agenda. Ways in which evidence can be misused include using evidence selectively, stifling the appropriate use of evidence, and creating of a spurious impression of uncertainty. The best way to detect and prevent the inappropriate use of evidence is to use processes that are systematic and transparent, as we will describe in subsequent articles in this series." (Source: "SUPPORT Tools for evidence-informed health Policymaking (STP)" Report from Norwegian Knowledge Centre for the Health Services No 4–2010, Chapter 1, p. 25. ISBN print 978-82-8121-313-5, ISBN digital 978-82-8121-334-0, ISSN 1890-1298)
"Conclusion
The SUPPORT tools in this series have been designed to help policymakers and those who support them to do one aspect of their job better or more efficiently – namely to find and use research evidence to support health policymaking. The tools are also relevant to health system stakeholders, such as non-governmental organisations and civil society groups. Different readers will use the tools in different ways. Policymakers may skim the articles to get ideas on how they should be adjusting the expectations they set for their staff. Those who support policymakers may choose to read a particular article to help them with undertaking a new activity, and then use the article later as a reference guide or as a way of refining their skills. We hope that policymakers and those who support them will help us to develop and improve what is presented here." (Source: ibid., p. 25)
Evidence-informed Health Policymaking (EIHP) Was Ignored
It is apparent NOKC's efforts and experience developing"SUPPORT Tools for evidence-informed health Policymaking (STP)" were not applied to Directorate of Health's knowledge-obviated, medically & ethically flawed 2014 Recommendation with their exclusive implementation of NCFM eSnurra Group's "method" (i.e., the appropriated, plagiarized, misused Hutchon Method of PDEE) within a government-mandated protocol of evidence-obviated medicine; a protocol proven to cause increased medical risks, critical medical mistakes and grievous medical harms with respect to obstetric medicine, fetal medicine and obstetric clinical care.
NOKC & Knowledge Brokering in Norway
Included below is a summary of NOKC from "Knowledge brokering in Norway: bringing rigour and transparency to policy inputs."
In summary, NOKC can be seen as an independent body that meets some, but not all, of the BRIDGE criteria for knowledge-brokering organizations (Chapter 2). On the one hand, NOKC:
http://www.euro.who.int/__data/assets/pdf_file/0017/270800/Bridge-Ch-8.pdf
https://www.ncbi.nlm.nih.gov/books/NBK458382/pdf/Bookshelf_NBK458382.pdf
https://www.ncbi.nlm.nih.gov/books/NBK458382/pdf/Bookshelf_NBK458382.pdf
https://www.ncbi.nlm.nih.gov/books/NBK458386/
http://www.euro.who.int/en/about-us/partners/observatory/publications/studies/bridging-the-worlds-of-research-and-policy-in-european-health-systems-2013
http://www.euro.who.int/en/about-us/partners/observatory/bridge-series
(Source: "Norway: Health system review. Health Systems in Transition" 2013; 15(8): 1–162. Ånen Ringard, Anna Sagan, Ingrid Sperre Saunes, Anne Karin Lindahl. European Observatory on Health Systems and Policies. ISSN 1817–6127 Vol. 15 No. 8)
http://www.euro.who.int/__data/assets/pdf_file/0018/237204/HiT-Norway.pdf
Included below is a summary of NOKC from "Knowledge brokering in Norway: bringing rigour and transparency to policy inputs."
In summary, NOKC can be seen as an independent body that meets some, but not all, of the BRIDGE criteria for knowledge-brokering organizations (Chapter 2). On the one hand, NOKC:
- gives policy-makers and some (if not all) stakeholders an explicit role in its governance and ensures they exercise their role with transparency and objectivity; 2 [2 This is complicated by the Directorate being not only one of the NOKC’s many policy clients but also the body to which it reports, whereas the Ministry and the broader set of organizations reporting to the Ministry (including the Directorate, regional health authorities and the Norwegian Medicines Agency) are the true clients that NOKC really serves in its policy-oriented work.] [
- has, and enforces, rules that ensure independence and address conflicts of interest;
- grants its director general the authority needed to ensure accountability to its knowledge-brokering mandate;
- is actively involved in networks that support its knowledge-brokering activities, including the Campbell Collaboration; Cochrane Collaboration; European Network for Health Technology Assessment (EUnetHTA); Guidelines International Network (G-I-N); Health Technology Assessment International (HTAi); and the International Network of Agencies for Health Technology Assessment (INAHTA) (and in some cases hosts their secretariats);
- collaborates with other knowledge-brokering organizations, both within the country and internationally; and
- establishes functional linkages with policy-making and stakeholder organizations.
- does not ensure an appropriate size, mix and capacity of staff with knowledge brokering responsibilities relative to its scale (the majority of staff being researchers and clinicians, a smaller number serving as administrators, and very few working as dedicated knowledge brokers on health systems and policy issues);
- does not ensure an appropriately diversified budget for knowledge brokering, with nearly complete budgetary dependence on the Norwegian government; and
- does not have an explicit approach to prioritizing knowledge brokering in general or urgent requests for knowledge syntheses in particular (the most frequent type of request), although it does have a formal process for making non-urgent requests for knowledge syntheses.
(Source: Govin Permanand, Anne Karin Lindahl, and John-Arne Røttingen (2013). "Knowledge brokering in Norway: bringing rigour and transparency to policy inputs." Chapter 8 in: Lavis JN, Catallo C and the BRIDGE Study Team, editors. Bridging the worlds of research and policy in European health systems. Copenhagen: WHO Regional Office for Europe on behalf of the European Observatory on Health Systems and Policies. ISBN 978 92 890 5029 6; Full NOKC version as PDF)
"NOKC is also the national and sole health technology assessment (HTA) agency in Norway, although hospitals will often undertake their own mini HTAs as a form of local decision support. NOKC is exploring ways to help facilitate the preparation of these mini HTAs." (Source: ibid., Ch. 8, p. 12)
http://www.euro.who.int/__data/assets/pdf_file/0017/270800/Bridge-Ch-8.pdf
https://www.ncbi.nlm.nih.gov/books/NBK458382/pdf/Bookshelf_NBK458382.pdf
https://www.ncbi.nlm.nih.gov/books/NBK458382/pdf/Bookshelf_NBK458382.pdf
https://www.ncbi.nlm.nih.gov/books/NBK458386/
http://www.euro.who.int/en/about-us/partners/observatory/publications/studies/bridging-the-worlds-of-research-and-policy-in-european-health-systems-2013
http://www.euro.who.int/en/about-us/partners/observatory/bridge-series
(Source: "Norway: Health system review. Health Systems in Transition" 2013; 15(8): 1–162. Ånen Ringard, Anna Sagan, Ingrid Sperre Saunes, Anne Karin Lindahl. European Observatory on Health Systems and Policies. ISSN 1817–6127 Vol. 15 No. 8)
http://www.euro.who.int/__data/assets/pdf_file/0018/237204/HiT-Norway.pdf