Ministry of Health & Care Services and Directorate of Health
Ministry of Health and Care Services sent their 20.11.2015 report: "National health and hospital plan (2016−2019)"
("Nasjonal helse- og sykehusplan (2016−2019)" to the Parliment (Storting).
Lailas case could have been a glowing example of that which Ministry of Health & Care Services envisioned and described in the excerpt below from Ministry of Health and Care Services' "National health and hospital plan (2016−2019)" which was sent to, and approved by, the Parliament (Storting) on 20.11.2015. Unfortunately, Directorate of Health's knowledge-obviated, medically & ethically flawed 2104 Recommendation, which Ministry of Health and Care Services approved, caused increased medical risks, critical medical mistakes and grievous medical harms to Laila and her baby, and other women and their fetuses/babies.
("Nasjonal helse- og sykehusplan (2016−2019)" to the Parliment (Storting).
Lailas case could have been a glowing example of that which Ministry of Health & Care Services envisioned and described in the excerpt below from Ministry of Health and Care Services' "National health and hospital plan (2016−2019)" which was sent to, and approved by, the Parliament (Storting) on 20.11.2015. Unfortunately, Directorate of Health's knowledge-obviated, medically & ethically flawed 2104 Recommendation, which Ministry of Health and Care Services approved, caused increased medical risks, critical medical mistakes and grievous medical harms to Laila and her baby, and other women and their fetuses/babies.
- "The patient role is changing. Patients increasingly request information about their own health and personal risk factors. Genetics and biotechnology are in rapid development, and individuals can now order advanced genetic research on the internet. Health service users expect both better quality and increased influence. New technology gives patients more responsibility and control. Creating the patient's health service causes changes for the healthcare staff. The patient's knowledge and experience will be used as a basis for improvement in the services, and healthcare professionals must regard the patient as an equal partner. If the possibilities for self-participation and participation are utilized in cooperation with the health service, it can lead to better health, less need for hospital attendance and fewer hospitalizations."
"Pasientrollen er i endring. Pasienter etterspør i større grad informasjon om egen helse og personlige risikofaktorer. Genetikk og bioteknologi er i rivende utvikling, og enkeltpersoner kan nå bestille avanserte genundersøkelser på internett. Brukere av helsetjenesten forventer både bedre kvalitet og økt innflytelse. Ny teknologi gir pasienter mer ansvar og kontroll. Å skape pasientens helsetjeneste medfører endringer for helsepersonellet. Pasientens kunnskap og erfaring skal brukes som grunnlag for forbedring i tjenestene, og helsepersonell må betrakte pasienten som en likeverdig samarbeidspartner. Dersom mulighetene for egeninnsats og medvirkning utnyttes i samarbeid med helsetjenesten, kan det føre til bedre helse, mindre behov for oppmøte på sykehuset og færre sykehusinnleggelser." (Source: "Meld. St. 11 (2015–2016) Melding til Stortinget: Nasjonal helse- og sykehusplan (2016–2019)" ("Meld. St. 11 (2015-2016) Message to the Storting: National Health and Hospital Plan (2016-2019)") 7 Den nye pasientrollen, p.55. Tilråding fra Helse- og omsorgsdepartementet 20. november 2015, godkjent i statsråd samme dag. (Regjeringen Solberg). (Recommendation from the Ministry of Health and Care Services November 20, 2015, approved by the State Council on the same day. (Solberg Government)))
Norway's Patients' Rights Act & Consent to Health Care
In consideration of Norway's Patient's Rights Act and consent to health care with respect to Laila's case, the facts articulated below are important.
Included below are Sections 4-1 & 4-2 of Chapter 4 of Norway's Patients' Rights Act [English] or Lov om pasient- og brukerrettigheter (pasient- og brukerrettighetsloven). These 2 Sections define patient consent. The Section headings for Sections 4.3 - 4.9 are also included for the purpose of demonstrating these Sections exist but do not apply to Laila's case.
Norway's Patients' Rights Act
Risk Mitigation in Obstetric & Fetal Medicine
Risk mitigation for women and their fetuses/babies in obstetric & fetal medicine requires an integrated, expert medical understanding and clinical competence in assessing both precautionary (fragility-based) and evidentiary (statistical-based) risk because the consequences of acting without an integrated understanding are too serious; and, this is precisely why protocols of obstetric medicine, fetal medicine and obstetric clinical care must remain within the domain of obstetric & fetal medicine experts, not Ministry politicians and not Directorate administrators whose respective competences of obstetric and fetal medicine are proven to be exceeded by even the most fundamental principle of pregnancy dating:
Additionally, in "Chapter 3. Specific duties and tasks" of Norway's "Act on Specialist Health Care (Special Health Services Act)" addresses the issues of "patient safety" and "causes of incidents to prevent similar happening again." However, and again, the problem is Directorate of Health's medically & ethically flawed 2014 Recommendation within a government-mandated protocol of evidence-obviated medicine that caused, and continues to cause, "substantial injury to the patient due to the provision of health services," as articulated in § 3.3. below. And, this next part is classic: "It should also be notified of incidents that could have led to serious injury." As we know, Directorate of Health were notified of an incident that not only could have led to serious injury, but did, and does, lead to serious injury; the"incident" was, of course, the implementation of Directorate of Health's medically & ethically flawed 2014 Recommendation against the clear, explicit, published warnings of the the risks and consequences by Norway's obstetric & fetal medicine experts, NGF and Bergen Group. And, while the prior point was classic, check out this next point: "...upon suspicion of a serious system failure shall notify the Directorate of Health Norwegian Board of Health." This "suspicion" qualification would obviate Laila's medical evidence, again, because LailasCase.com is fact-based evidence of the actual, real-world systemic risks and consequences of which NGF and Bergen Group had clearly, explicitly and publicly warned Directorate of Health 2 years before Laila became pregnant. Consequently, LailasCase.com would have to be obviated from consideration because LailasCase.com is fact-based evidence, not a "suspicion." The law states: "suspicion," not fact-based evidence. Sound familiar? Now, consider this closing point: "With serious incident to mean death or significant damage to the patient in whom the outcome is unexpected in relation to foreseeable risk." Again, and while Laila did have an unnecessary, unwanted Cesarean section surgery with a cascade of complications, and her baby endured an unidentified, prolonged, undiagnosed, untreated fetal growth restriction/malformation of her head, this did not constitute a "serious incident" because the associated increased medical risks, critical mistakes and grievous medical harms were not "unexpected" even though there was "significant damage to the patient." So, did you catch it? Of course you did. All the risks and consequences of Laila's case had been "foreseeable risks" because the risks and consequences had been foreseen by NGF and Bergen Group who had warned Directorate of Health of same more than 2 years before Laila became pregnant. Moreover, Laila & Edward had warned of the risks and consequences of using a grossly inaccurate NCFM eSnurra BPD-based EDD, including the masking of fetal pathology, since Laila's routine 18wUSW, the last warning Laila & Edward issued was 15-hours before Laila went into labor. Therefore, all the risks in Laila's case had been foreseen 2 years before Laila's pregnancy and, separately, by Laila & Edward during Laila's pregnancy. The, law states if the risks were "foreseeable," then there was no "serious incident" and, consequently, no harm, no foul. All that which is quoted above is in red bold text below. And, even though the last excerpt below, § 3.4 a., is not quoted, it is a must read.
In consideration of Norway's Patient's Rights Act and consent to health care with respect to Laila's case, the facts articulated below are important.
- Laila had been told the purpose of the routine 18-week ultrasound exam was to ensure there were no observable problems with the anatomy and position of her baby. There was no mention of ultrasound-based dating of her pregnancy.
- Laila never gave consent, implicitly, explicitly, tacitly or otherwise, to have her key, factual pregnancy dates (i.e., LMPD/OTPD/SCID) obviated from medical evidence without her prior, informed voluntary, explicit consent, and replaced with an ultrasound-based estimate of EDD and, therefrom, a calculated GA, using the equivalent of Naegele's rule, in reverse, to be used, unilaterally, in all medical thinking, medical decision-making and medical actions for the duration of her pregnancy.
- Laila never received the necessary information concerning "the content of the health care," i.e., the full purpose and hidden conditions of the routine 18-week ultrasound exam conducted by the specialist services using NCFM eSnurra Group's "method" within a government-mandated protocol of evidence-obviated medicine. This was never disclosed.
- Laila was never informed of the risks and, specifically, that there was a 12.8% risk that the NCFM eSnurra BPD-based EDD & GA could be grossly inaccurate (i.e. EDD more than 14 days early or late, and GA a couple of days less); a risk no rational, informed pregnant woman would assume for herself and her fetus/baby if she were to be in ownership possession of a factual LMPD/OTPD/SCID, or an OTPD, or a SCID, or just a reliable LMPD given there is just a 6% risk ultrasound will be more accurate than a reliable LMPD, i.e., as published by NCFM Snurra Group's Tunón et al. 1996.
Included below are Sections 4-1 & 4-2 of Chapter 4 of Norway's Patients' Rights Act [English] or Lov om pasient- og brukerrettigheter (pasient- og brukerrettighetsloven). These 2 Sections define patient consent. The Section headings for Sections 4.3 - 4.9 are also included for the purpose of demonstrating these Sections exist but do not apply to Laila's case.
Norway's Patients' Rights Act
- Chapter 4. Consent to health care
Section 4-1. General rule relating to consent Health care may only be provided with the patient’s consent, unless legal authority exists or there are other valid legal grounds for providing health care without consent. In order for the consent to be valid, the patient must have received the necessary information concerning his health condition and the content of the health care.
The patient may withdraw his consent. If the patient withdraws consent, the health care provider shall give the necessary information regarding the consequences of not giving the health care.
Section 4-2. Requirements regarding the form of consent
Consent may be given explicitly or tacitly. Tacit consent is considered to have been given if it is probable, based on the patient’s conduct and all other circumstances, that he or she accepts the health care.
The Ministry may issue regulations regarding a requirement of written consent or other formal requirements in connection with certain types of health care.
Section 4-3. Who has competence to give consent
Section 4-4. Consent on behalf of children
Section 4-5. Consent on behalf of young people who are not competent to give consent
Section 4-6. Consent on behalf of persons who are of full legal age and legal capacity and who are not competent to give consent
Section 4-7. Patients who have been declared legally incapacitated
Section 4-8. Patients who are not competent to give consent and who have no next of kin
Section 4-9. The patient’s right to refuse health care in special situations
(Source: The Act of 2 July 1999 No. 63 relating to Patients’ Rights (Patients’ Rights Act [English] or Lov om pasient- og brukerrettigheter)
Risk Mitigation in Obstetric & Fetal Medicine
Risk mitigation for women and their fetuses/babies in obstetric & fetal medicine requires an integrated, expert medical understanding and clinical competence in assessing both precautionary (fragility-based) and evidentiary (statistical-based) risk because the consequences of acting without an integrated understanding are too serious; and, this is precisely why protocols of obstetric medicine, fetal medicine and obstetric clinical care must remain within the domain of obstetric & fetal medicine experts, not Ministry politicians and not Directorate administrators whose respective competences of obstetric and fetal medicine are proven to be exceeded by even the most fundamental principle of pregnancy dating:
- "the basis for determining gestational age is not the end of the pregnancy, but its beginning."
(Source: "Flawed recommendation issued by the Norwegian Directorate of Health concerning the determination of fetal age" or "Helsedirektoratet gir feil anbefaling om bestemmelse av fosteralder" Cathrine Ebbing, MD, PhD, Synnøve Lian Johnsen MD, PhD, Jørg Kessler, MD, PhD, Torvid Kiserud, MD, PhD, Svein Rasmussen, MD, PhD., Nr. 8, 5 mai 2015, Tidsskr Nor Legeforen, 2015; 135:7401, DOI: 10.4045/tidsskr.15.0093) [Note: Fragility-based risk
- Chapter 2. Responsibility and general tasks
§ 1.2. State responsibility for specialist
The State has the overall responsibility for ensuring that the population is offered the necessary specialist.
0 Added by Act of 15 June 2001 No.. 93 (ikr. January 1, 2002 acc. Res. 14 Dec 2001 no. 1417).
(Source: https://lovdata.no/dokument/NL/lov/1999-07-02-61)
Additionally, in "Chapter 3. Specific duties and tasks" of Norway's "Act on Specialist Health Care (Special Health Services Act)" addresses the issues of "patient safety" and "causes of incidents to prevent similar happening again." However, and again, the problem is Directorate of Health's medically & ethically flawed 2014 Recommendation within a government-mandated protocol of evidence-obviated medicine that caused, and continues to cause, "substantial injury to the patient due to the provision of health services," as articulated in § 3.3. below. And, this next part is classic: "It should also be notified of incidents that could have led to serious injury." As we know, Directorate of Health were notified of an incident that not only could have led to serious injury, but did, and does, lead to serious injury; the"incident" was, of course, the implementation of Directorate of Health's medically & ethically flawed 2014 Recommendation against the clear, explicit, published warnings of the the risks and consequences by Norway's obstetric & fetal medicine experts, NGF and Bergen Group. And, while the prior point was classic, check out this next point: "...upon suspicion of a serious system failure shall notify the Directorate of Health Norwegian Board of Health." This "suspicion" qualification would obviate Laila's medical evidence, again, because LailasCase.com is fact-based evidence of the actual, real-world systemic risks and consequences of which NGF and Bergen Group had clearly, explicitly and publicly warned Directorate of Health 2 years before Laila became pregnant. Consequently, LailasCase.com would have to be obviated from consideration because LailasCase.com is fact-based evidence, not a "suspicion." The law states: "suspicion," not fact-based evidence. Sound familiar? Now, consider this closing point: "With serious incident to mean death or significant damage to the patient in whom the outcome is unexpected in relation to foreseeable risk." Again, and while Laila did have an unnecessary, unwanted Cesarean section surgery with a cascade of complications, and her baby endured an unidentified, prolonged, undiagnosed, untreated fetal growth restriction/malformation of her head, this did not constitute a "serious incident" because the associated increased medical risks, critical mistakes and grievous medical harms were not "unexpected" even though there was "significant damage to the patient." So, did you catch it? Of course you did. All the risks and consequences of Laila's case had been "foreseeable risks" because the risks and consequences had been foreseen by NGF and Bergen Group who had warned Directorate of Health of same more than 2 years before Laila became pregnant. Moreover, Laila & Edward had warned of the risks and consequences of using a grossly inaccurate NCFM eSnurra BPD-based EDD, including the masking of fetal pathology, since Laila's routine 18wUSW, the last warning Laila & Edward issued was 15-hours before Laila went into labor. Therefore, all the risks in Laila's case had been foreseen 2 years before Laila's pregnancy and, separately, by Laila & Edward during Laila's pregnancy. The, law states if the risks were "foreseeable," then there was no "serious incident" and, consequently, no harm, no foul. All that which is quoted above is in red bold text below. And, even though the last excerpt below, § 3.4 a., is not quoted, it is a must read.
- Norway's Act on Specialist Health Care (Special Health Services Act)
§ 3.3. Duty to report to the Health Directorate
The purpose of the notification requirement is to improve patient safety. The messages will be used to clarify the causes of incidents and to prevent similar happening again.
Health institutions covered by this Act shall, notwithstanding the confidentiality immediately notify the Directorate of Health about substantial injury to the patient due to the provision of health services or by a patient injuring another. It should also be notified of incidents that could have led to serious injury. The notification shall not be directly personally identifiable information.
Health Directorate shall treat the messages to build up and disseminate knowledge to health professionals, health service users, responsible authorities and manufacturers on measures that can be implemented to improve patient safety.
Health Directorate shall ensure that information about individuals can not be attributed to the person concerned.
Upon suspicion of a serious system failure shall notify the Directorate of Health Norwegian Board of Health.
Message to the Directorate of Health can not in itself form the basis for initiating action or make decisions on reaction pursuant to the Health Chapter 11. The same applies to a request for prosecution by the Health § 67, second paragraph.
Ministry may issue further provisions regarding the messages mentioned in the second paragraph, including the content of your messages. The Ministry may decide that the notification requirement shall also apply for specialist that is not covered by the second paragraph.
0 Amended by laws 29 August 2003 No.. 87 (ikr. September 1, 2003 acc. Res. 29 August 2003 No.. 1092), 24 June 2011 No.. 30 (ikr. July 1, 2012 acc. Res. 16 Dec 2011 No.. 1252) as amended by Act 22 June 2012 No.. 46, 18 Dec 2015 No.. 121 (ikr. January 1, 2016).
§ 3-3 a. Notification of the National Board of Health about serious incidents
To ensure supervisory review health authorities and businesses that deal with health or regional health authorities should immediately notify the Board of Health about serious incidents. With serious incident meant death or significant damage to the patient in whom the outcome is unexpected in relation to foreseeable risk.
The Ministry may issue further provisions on notification under the first paragraph, including the contents of the notifications. The Ministry may issue regulations also determine that other healthcare than those stated in the first paragraph shall be covered by the notification requirement.
0 Added by Act of 24 June 2011 No.. 30 (ikr. January 1, 2012 acc. Res. 16 Dec 2011 No.. 1252). Amended by Act of June 16, 2017 no. 56 (ikr. From the King decides).
§ 3.4. Quality and patient selection
Health institutions covered by this Act, shall establish quality and patient selection as part of the systematic work institution shall perform in accordance with § 2-1, third paragraph, and § 3-4 a.
The committees may notwithstanding the confidentiality require information necessary for carrying out their work.
The Ministry may issue more detailed provisions on the committees' work, including whether the information to patients and the impartiality of its members.
0 Amended by Act of 24 June 2011 No.. 30 (ikr. January 1, 2012 acc. Res. 16 Dec 2011 No.. 1252).
§ 3.4 a. Quality improvement and patient
Anyone who provides health services under this Act shall ensure that the enterprise is working systematically to improve quality and patient safety.
The Ministry may issue further provisions regarding the obligation under the first paragraph.
0 Added by Act of 24 June 2011 No.. 30 (ikr. January 1, 2012 acc. Res. 16 Dec 2011 No.. 1252).
(Source: https://lovdata.no/dokument/NL/lov/1999-07-02-61)
The Norwegian Health Care System
(Source: "International Profiles of Health Care Systems" Australia, Canada, China, Denmark, England, France, Germany, India, Israel, Italy, Japan, the Netherlands, New Zealand, Norway, Singapore, Sweden, Switzerland, Taiwan, and the United States. MAY 2017. EDITED BY Elias Mossialos and Ana Djordjevic, London School of Economics and Political Science, Robin Osborn and Dana Sarnak, The Commonwealth Fund) [Note: On 01 January of 2016, Ministry of Health and Care Services separated Norwegian Knowledge Centre for the Health Services (Nasjonalt kunnskapssenter for helsetjenesten) (NOKC) from Norwegian Directorate of Health to merge NOKC into the Norwegian Institute of Public Health (NIPH). Since then, NOKC has been known as NIPH Division of Health Services.]
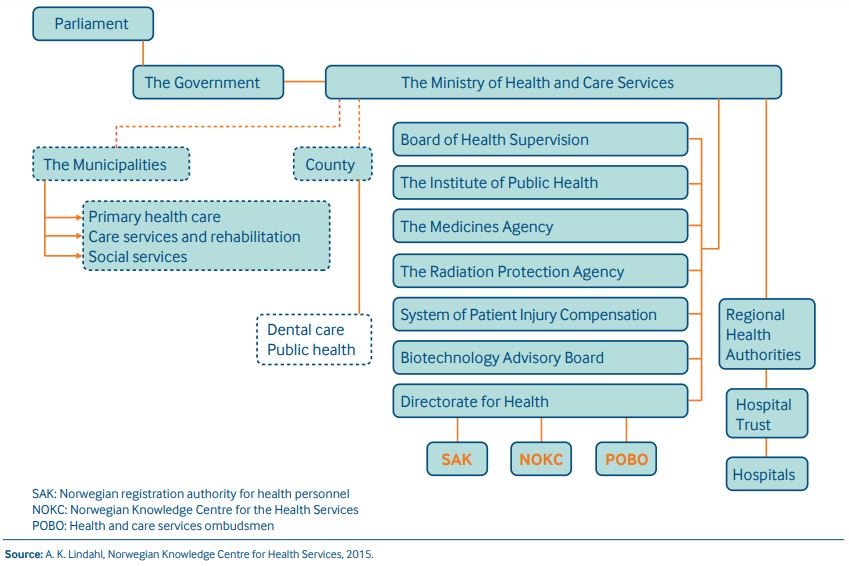
The Big-picture View: Ministry of Health and Care Services
Included below is the "Management Hierarchy" of Norwegian Ministry of Health and Care Services (Helse- og omsorgsdirektoratatet) as presented by Norwegian Center for Research Data (NSD) (Norsk senter for forskningsdata). This "Management Hierarchy" is included to provide a big-picture view. Also, the NSD website is worth browsing.
(Source: http://www.nsd.uib.no/polsys/data/en/forvaltning/enhet/39000/hierarki)
• The State (Norway)
Included below is the "Management Hierarchy" of Norwegian Ministry of Health and Care Services (Helse- og omsorgsdirektoratatet) as presented by Norwegian Center for Research Data (NSD) (Norsk senter for forskningsdata). This "Management Hierarchy" is included to provide a big-picture view. Also, the NSD website is worth browsing.
(Source: http://www.nsd.uib.no/polsys/data/en/forvaltning/enhet/39000/hierarki)
• The State (Norway)
- •The Storting (unicameral legislature)
- •The Government
- Ministry of Health and Care Services
- * Administrative Department
- •* Budget and Economic Department
- •* Health law department (HRA)
- * Local authority services department
- •* Ownership department (EIA)
- * Public health department
- * Specialist heath services department
- •Communications Division
- •Department of eHealth
- Subordinate units:
- •* Directorate for e-health
- •Norwegian Board of Health Supervision
- •Norwegian Medicines Agency
- •The Norwegian Biotechnology Advisory Board
- •* National administrative appeals body for health services
- Norwegian Directorate of Health
- Norwegian Institute of Public Health (NIPH)
- •Norwegian System of Compensation to Patients
- Affiliated companies:
- •AS Vinmonopolet
- •* Norwegian Healthnet - Public Enterprise
- •* Health service organization for emergency network HF
- •* Hospital Build HF
- •Central Norway Regional Health Authoritiy
- •National ICT
- •Northern Norway Regional Health Authority
- Southern and Eastern Norway Regional Health Authority
- Affiliated foundations:
- •FRAMBU - Centre for Rare Disorders
- •Western Norway Regional Health Authority
- Affiliated foundations:
- •* Beitostølen health-building sport centre
- •* Motebello centre
- •* Valnesfjord health-building sport centre
- •Institute for Cancer Research, the Norwegian Radium Hospital
- •The Radium Hospital Research Foundation
- Ministry of Health and Care Services
- •The Government
Included below is the "Management Hierarchy" of Norwegian Directorate of Health, as presented by Norwegian Center for Research Data (NSD) (Norsk senter for forskningsdata). Again, this is included to provide a big-picture understanding. (Source: http://www.nsd.uib.no/polsys/data/en/forvaltning/enhet/37800/hierarki)
The State (Norway)
The State (Norway)
- •The Storting (unicameral legislature)
Ministry of Health and Care Services' "Norwegian Patient Safety Programme: In Safe Hands"
Included below is the introduction to Ministry of Health and Care Services' "Norwegian Patient Safety Programme: In Safe Hands" The purpose of this program is to reduce patient harm and improve patient safety. Red bold text is used to highlight the point that patient safety and patient harms are identified problems; problems for which a 20-member Steering Committee (Styringsgruppen) was assembled. Moreover, the red bold text serves as evidence of much talk-the-talk competence.
The Norwegian Patient Safety Programme: In Safe Hands
In Safe Hands was originally launched in 2011 as a patient safety campaign by the Norwegian Ministry of Health and Care Services. From 2014, the campaign continued as a five-year programme. The aim of the programme is to reduce patient harm.
Global Trigger Tool (GTT)
The tool referenced above "Global Trigger Tool" evokes a military theme. One cannot help noticing the similarities between military and healthcare lexicons for transitioning from harms & deaths to more palatable terms and, then, to convenient, innocuous abbreviations.
Ministry of Health and Care Services approved Directorate of Health's knowledge-obviated, medically & ethically flawed 2014 Recommendation with their exclusive implementation of NCFM eSnurra Group's "method" (i.e., the appropriated, plagiarized, misused Hutchon Method of PDEE) within a government-mandated protocol of evidence-obviated medicine; a protocol proven to cause increased medical risks (adverse events or AEs), critical medical mistakes (also adverse events or AEs) and grievous medical harms (again, also adverse events or AEs) to some of Norway's women (patients) and their fetuses/babies (also patients). The statement from Ministry of Health and Care Services which speaks most directly to this insidious systemic problem is:
Incident reporting – a difficult balancing act
"The purpose of reporting adverse incidents is not to point to scapegoats, but to increase patient safety. Nevertheless, many professionals fail to report unwanted incidents, a study shows."
Norwegian Board of Health Supervision
"The Norwegian Board of Health Supervision is a national public institution organized under the Ministry of Health and Care Services, with responsibility for supervision of child welfare services, social services, and health and care services." (Source: Norwegian Board of Health Supervision website: https://www.helsetilsynet.no/Norwegian-Board-of-Health-Supervision/)
In February 2017 Norwegian Board of Health Supervision’s Investigation Unit for Serious and Adverse Events published: "Inconceivable or predictable? Follow-up of notifications of serious events in the specialist health service" Status and experience 2016 from the Surveillance Unit of the Norwegian Board of Health" ("Utenkelig eller forutsigbart? Oppfølging av varsler om alvorlige hendelser i spesialisthelsetjenesten" Status og erfaringer 2016 fra Undersøkelsesenheten i Statens helsetilsyn).
"The report has two purposes: (Rapporten har to formål:)
First, two approbations for Norwegian Board of Health Supervision (Statens helsetilsyn).
In March 2014, Norwegian Board of Health Supervision’s Investigation Unit for Serious and Adverse Events [in in the Specialist Health Services] published: "Could this have happened here? Examples and experience gained from investigation of serious adverse events 2010–2013" REPORT OF THE NORWEGIAN BOARD OF HEALTH SUPERVISION 3/2014. However, the Norwegian title of this publication is more descriptive, despite the apparent exclusion of the words "and adverse."
Included below is the introduction to Ministry of Health and Care Services' "Norwegian Patient Safety Programme: In Safe Hands" The purpose of this program is to reduce patient harm and improve patient safety. Red bold text is used to highlight the point that patient safety and patient harms are identified problems; problems for which a 20-member Steering Committee (Styringsgruppen) was assembled. Moreover, the red bold text serves as evidence of much talk-the-talk competence.
The Norwegian Patient Safety Programme: In Safe Hands
In Safe Hands was originally launched in 2011 as a patient safety campaign by the Norwegian Ministry of Health and Care Services. From 2014, the campaign continued as a five-year programme. The aim of the programme is to reduce patient harm.
- Goals and objectives
The overall aim of In Safe Hands is to reduce patient harm and improve patient safety in Norway. To accomplish this, three objectives have been defined:- Reduce preventable patient harm.
- Establish lasting structures for patient safety.
- Improve the patient safety culture in the health and care services.(Source:
- Patient Safety Culture Measurement
Publisert 21.05.2012 09.57 Oppdatert 09.11.2016 12.42
The “In Safe Hands” campaign is currently measuring patient safety culture among professionals in all care giving units in every hospital. In addition, the campaign is now piloting a similar survey on patient safety culture among a selection of ambulatory clinics with regular general practitioners and emergency care. - What is patient safety culture?
The notion of culture implies ”the way we do things here”. The patient safety culture describes to what extent the professional working environment is characterized by routines and priorities that prevent adverse events and patient harm. - How important is patient safety culture?
Patient safety culture is very important when it comes to explaining the variety in adverse events and patient harm between different care giving units and hospitals. All employees in healthcare services wish to avoid adverse events, however sometimes established routines and practices represent a threat to patient safety. First and foremost it is a question to what degree a clinic prioritizes and values safe patient treatment, for instance by giving positive feedback to employees that need assistance, or by ensuring that professionals have the necessary competence for the procedures they are set to do. It is also a matter of to which extent a clinic practices routines for learning from adverse events. - How do we measure patient safety culture?
We are measuring patient safety culture by asking all employees involved in the treatment of patients about how they feel that adverse events and patient harm is handled and discussed in the workplace environment. For the survey we use a Norwegian version of Safety Attitudes Questionnaire – SAQ, which has been used in patient safety culture measurements in several other countries.
How do we carry out the survey?
All employees with direct patient contact receive an e-mail with an electronic version of the questionnaire. The survey treats every respondent anonymously, and it takes around 15 minutes to complete the questionnaire. We aim at a 70 per cent response rate.
What happens next?
The measuring of patient safety culture will provide hospitals and clinics with more knowledge about how the employees experience that their healthcare institution works to reduce the patient harm. (Source: "The Norwegian Patient Safety Programme: In Safe Hands" Publisert 12.08.2011 12.21 Oppdatert 09.11.2016 12.43)
http://www.pasientsikkerhetsprogrammet.no/om-oss/english/the-norwegian-patient-safety-programme-in-safe-hands
Global Trigger Tool (GTT)
The tool referenced above "Global Trigger Tool" evokes a military theme. One cannot help noticing the similarities between military and healthcare lexicons for transitioning from harms & deaths to more palatable terms and, then, to convenient, innocuous abbreviations.
- Military: civilian (non-combatant) deaths & injuries > collateral damage > CD
- Healthcare: patient harms & deaths > adverse events > AE
- IHI Global Trigger Tool for Measuring Adverse Events
Institute for Healthcare Improvement (IHI)
Cambridge, Massachusetts, USA
(Source: http://www.ihi.org/resources/Pages/Tools/IHIGlobalTriggerToolforMeasuringAEs.aspx)
"The use of "triggers," or clues, to identify adverse events (AEs) is an effective method for measuring the overall level of harm in a health care organization. The IHI Global Trigger Tool for Measuring AEs provides instructions for training reviewers in this methodology and conducting a retrospective review of patient records using triggers to identify possible AEs. This tool includes a list of known AE triggers as well as instructions for selecting records, training information, and appendices with references and common questions. The tool provides instructions and forms for collecting the data you need to track three measures:
· Adverse Events per 1,000 Patient Days
· Adverse Events per 100 Admissions
· Percent of Admissions with an Adverse Event
For more general information on Trigger Tools and how to select the appropriate one, see the Introduction to Trigger Tools page."
Background
"Traditional efforts to detect AEs have focused on voluntary reporting and tracking of errors. However, public health researchers have established that only 10 to 20 percent of errors are ever reported and, of those, 90 to 95 percent cause no harm to patients. Hospitals need a more effective way to identify events that do cause harm to patients, in order to select and test changes to reduce harm. In 2000, a group of IHI faculty consisting of clinical experts and other professionals developed the first IHI Trigger Tool in an effort to detect a greater number of AEs. Over time, multiple topic and location specific Trigger Tools have been developed and the IHI Global Trigger Tool combines several of these into one tool that can be used to measure harm at the hospital level." (Source: ibid.)
Ministry of Health and Care Services approved Directorate of Health's knowledge-obviated, medically & ethically flawed 2014 Recommendation with their exclusive implementation of NCFM eSnurra Group's "method" (i.e., the appropriated, plagiarized, misused Hutchon Method of PDEE) within a government-mandated protocol of evidence-obviated medicine; a protocol proven to cause increased medical risks (adverse events or AEs), critical medical mistakes (also adverse events or AEs) and grievous medical harms (again, also adverse events or AEs) to some of Norway's women (patients) and their fetuses/babies (also patients). The statement from Ministry of Health and Care Services which speaks most directly to this insidious systemic problem is:
- "All employees in healthcare services wish to avoid adverse events, however sometimes established routines and practices represent a threat to patient safety."
- the government, their employer, via Directorate of Health's medically & ethically flawed government-mandated protocol of evidence-obviated medicine, or
- the patient(s), with the evidence of their own senses, education, training, knowledge and experience in the practice of evidence-based medicine
- advocate for Directorate of Health's medically & ethically flawed national medical policy with their government-mandated protocol of evidence-obviated medicine even when confronted with fact-based medical evidence of known, proven efficacy which contradicts the mandated protocol; a mandated protocol which causes increased medical risks, critical medial mistakes and grievous medical harms (including perinatal death), unnecessarily.
- advocate for patients' best medical interests and safety first and foremost, by considering all available information in the practice of evidence-based medicine.
- "DOUBLETHINK means the power of holding two contradictory beliefs in one’s mind simultaneously, and accepting both of them. The Party intellectual knows in which direction his memories must be altered; he therefore knows that he is playing tricks with reality; but by the exercise of DOUBLETHINK he also satisfies himself that reality is not violated. The process has to be conscious, or it would not be carried out with sufficient precision, but it also has to be unconscious, or it would bring with it a feeling of falsity and hence of guilt" (Source: George Orwell, "1984," Part Two, Chapter 9)
- "Lastly a word on the centrality of the patient in any discussion on medical ethics. Most medical associations acknowledge in their foundational policies that ethically, the best interests of the individual patient should be the first consideration in any decision on care. This course will only succeed if it amplifies this imperative – TO PUT THE PATIENT FIRST." (Source: "Course description Medical Ethics")
The "Fundamentals of Medical Ethics" was authored by John R. Williams, former Director of Ethics at the World Medical Association (WMA). Interestingly, Torunn Janbu, Director of Hospital Services of the Specialized Health Care Services, Norwegian Directorate of Health was President, Norwegian Medical Association (2005-2007) and, former Chairperson, Medical Ethics Committee, WMA (2010-2013). Consequently, one can only wonder how the wheels came off Torunn Janbu's medical ethics wagon with respect to her leadership role in promoting, deciding and implementing Directorate of Health's knowledge-obviated, medically & ethically flawed 2014 Recommendation; a Recommendation that implemented a protocol of evidence-obviated medicine; a Recommendation which serves as a textbook example of systemic, harms-causing research misconduct, the bending of policy-relevant science, manipulation and corruption.
- "It is also a matter of to which extent a clinic practices routines for learning from adverse events."
Incident reporting – a difficult balancing act
"The purpose of reporting adverse incidents is not to point to scapegoats, but to increase patient safety. Nevertheless, many professionals fail to report unwanted incidents, a study shows."
- ABSTRACT
Background: Although health authorities expect patient safety incidents to be reported, research shows that such incidents are underreported, and the reasons for this are currently being debated. Knowledge is needed on the factors that promote and inhibit incident reporting in the health services.
Objectives: To describe factors that influence midwives’ and mental health nurses’ decisions on whether or not to report adverse incidents.
Method: The study has a quantitative design. Data were collected using a self-administered written questionnaire. The respondents were midwives and mental health nurses (n = 133).
Main results: The majority of informants were familiar with the procedures for reporting adverse incidents, but had more knowledge of the procedures for reporting clinical errors than unethical practice. The main motivation for reporting such incidents was to avoid harm to patients. However, the informants’ own interests or the interests of their work colleagues affected the decision of whether or not to report.
Conclusion: The midwives and mental health nurses highly prioritised the patients’ rights. Still, reporting adverse incidents is complicated and is influenced by cultural and organisational factors. (Source: "Incident reporting – a difficult balancing act" Ingeborg Ulvund, Høgskolelektor, Avd. for helse- og sosialfag, Høgskolen i Molde; Anne-Marie Mork Rokstad, Førsteamanuensis II Avd. for helse- og sosialfag, Høgskolen i Molde; Solfrid Vatne Professor, Avd. for helse- og sosialfag, Høgskolen i Molde. Sykepleie, utgis av Norsk Sykepleierforbund. PEER-REVIEWED RESEARCH PUBLISHED: 27.01.2017 UPDATED: 27.01.2017. Original pdf: https://sykepleien.no/sites/default/files/pdf-export/pdf-export-58904_0.pdf)
Norwegian Board of Health Supervision
"The Norwegian Board of Health Supervision is a national public institution organized under the Ministry of Health and Care Services, with responsibility for supervision of child welfare services, social services, and health and care services." (Source: Norwegian Board of Health Supervision website: https://www.helsetilsynet.no/Norwegian-Board-of-Health-Supervision/)
In February 2017 Norwegian Board of Health Supervision’s Investigation Unit for Serious and Adverse Events published: "Inconceivable or predictable? Follow-up of notifications of serious events in the specialist health service" Status and experience 2016 from the Surveillance Unit of the Norwegian Board of Health" ("Utenkelig eller forutsigbart? Oppfølging av varsler om alvorlige hendelser i spesialisthelsetjenesten" Status og erfaringer 2016 fra Undersøkelsesenheten i Statens helsetilsyn).
- "The Unit investigates serious adverse events reported by hospitals, in some cases by visiting the hospital where the event happened. Patients and relatives are given the right to receive information." (Source: Norwegian Board of Health website: https://www.helsetilsynet.no/Norwegian-Board-of-Health-Supervision/).
"The report has two purposes: (Rapporten har to formål:)
- To contribute to analysis and reflection in health enterprises based on the notifications (learning perspective)
(å bidra til analyse og refleksjon i helseforetakene med utgangspunkt i varslene (læringsperspektiv)) - Reporting alerts and notification management in the form of statistics / activity figures"
(å rapportere om varsler og varselhåndteringen i form av statistikk/aktivitetstall)
First, two approbations for Norwegian Board of Health Supervision (Statens helsetilsyn).
- Kudos for the metaphorical black swan on the cover. (See: Black swan theory)
- Double kudos for citing and referencing Nassim Nicholas Taleb, the developer of black swan theory, whose motto, as stated on his website's Home Page, is:"If you see fraud and don't shout fraud, you are a fraud." Taleb's motto is quoted in the footer of each page of LailasCase.com.
- Laila could go into labor, under the umbrella or normal variance for her factual LMPD/OTPD/SCID-based GA & EDD before her baby had been turned from breech to vertex for normal delivery. This was an easily "computeable" probability, with known risk and consequences, i.e., an unwanted, unnecessary higher-risk breech delivery or Cesarean section surgery delivery, whereas a tenant of a black swan event is the "non-computability" of probability.
- Laila's baby would be born with the effects of a fetal growth restriction/malformation of her head, or worse.
In March 2014, Norwegian Board of Health Supervision’s Investigation Unit for Serious and Adverse Events [in in the Specialist Health Services] published: "Could this have happened here? Examples and experience gained from investigation of serious adverse events 2010–2013" REPORT OF THE NORWEGIAN BOARD OF HEALTH SUPERVISION 3/2014. However, the Norwegian title of this publication is more descriptive, despite the apparent exclusion of the words "and adverse."
- "Svikt i samhandling, kommunikasjon og kompetanse i alvorlige hendelser …… kunne det skjedd hos oss? Eksempler og erfaringer 2010–2013 fra Undersøkelsesenhetens arbeid med varsler om alvorlige hendelser i spesialisthelsetjenesten (§ 3-3a i spesialisthelsetjenesteloven) (Rapport fra Helsetilsynet 3/2014)"
Failure of interaction, communication and competence in serious events ...... could it happen to us? Examples and experiences 2010-2013 from the Surveillance Unit's work on notifications of serious events in the specialist health service (Section 3-3a of the Specialist Health Services Act) (Report from the Norwegian Board of Health and Safety 3/2014)
- "In a society built on welfare, services must have broad backing in the general public. If patients are to have trust in the delivered services, these must be reliable and of high quality. A key premise for trust is that patients and others perceive the delivered services as being of high quality and reliable. Sound supervision of the services helps foster trust."
- "Unexpected and serious adverse events are about people, and about hospitals and health personnel who want to do a good job ̶but who sometimes fall short. However, first and foremost such events are about patients in need of medical treatment, and about their immediate family. It is the experience of the Norwegian Board of Health Supervision that relatives often have relevant and solid information about what has happened – information that supplements and adds new layers of understanding to the accounts given by health personnel and their leaders. Combining information from next-of-kin with that supplied by staff thus produces a comprehensive picture encompassing as many aspects of the events as possible."
Breach of Medical Ethics
The breaches of medical ethics inherent in Directorate of Health's medically & ethically flawed 2014 Recommendation seem too numerous to list. Included below are excerpts from the Norwegian Medical Association (Den norske legeforening) website "Code of Ethics for Doctors" "Adopted by the Representative Body in 1961 and subsequently amended, most recently in 2015." All text in bold font is from "Code of Ethics for Doctors" with comments following in plain text. Needless to say, a medical ethics expert would have a field day and, probably, a nice paper to publish, by analyzing Directorate of Health's medically & ethically flawed 2014 Recommendation. [Note: Yes, that was an invitation to medical ethics experts who talk the talk and walk the walk.] (Source: http://legeforeningen.no/om-legeforeningen/organisasjonen/rad-og-utvalg/organisasjonspolitiske-utvalg/etikk/code-of-ethics-for-doctors-/)
American College of Physicians Ethics Manual: Sixth Edition
Included below is an excerpt from the American College of Physicians Ethics Manual: Sixth Edition. The entirety of this manual is available via the links included below. It is interesting to note the ACP Ethics Manual has 43 occurrences of the word "consent" and 16 occurrences of the 2-word sequence "informed consent."
The Biotechnology Act: Excluded the Routine 18-Week Ultrasound Exam
Intentional Recklessness & Willful Negligence
Norway's experts in obstetric medicine, fetal medicine and obstetric clinical care were:
Directorate of Health's 2014 Recommendation was formulated, decided and implemented in an intentionally substandard fashion, the consequences of which included increased medical risks, critical medical mistakes and grievous medical harms of which Directorate of Health were clearly and explicitly warned by NGF, Bergen Group and others.
The following excerpts articulate the foundation upon which Directorate of Health claimed to have based their medically & ethically flawed 2014 Recommendation.
NOKC's Reply to Directorate of Health or "The Fix"
Included below is a translation of the reply NOKC sent to Directorate of Health. NOKC's reply is included in its entirety due to the significance of what NOKC states and, more importantly, what NOKC does not state. URL links have been added to the the Case number and the 5 cited references for ... wait for it ... easy reference.
Norway's FOIA Exemptions: A Potential Shield for Corruption & Willful Negligence?
The many exemptions in Norway's Freedom of Information Act (offentleglova) can be used to shield Directorate of Health's: unethical behavior, breach of medical ethics, professional misconduct, conflict of interest, acting beyond competence, actively ignoring warnings of the risks and consequences from Norway's medical experts, thereby, engaging in intentional recklessness, willful negligence, and, possibly, criminal activity that served to corrupt Directorate of Health's:
FOIA Exemption: Internal Documents
The interest of public access to information, especially regarding the formulation and decision-making processes of national healthcare policy and associated implementation protocols, must outweigh the need for FOIA exemption. For reference, Norway's Freedom of Information Act (FOIA) is known as Lov om rett til innsyn i dokument i offentleg verksemd (offentleglova) which is also known as: "Act of 19 May 2006 No. 16 relating to the right of access to documents held by public authorities and public undertakings (short title: Freedom of Information Act (offentleglova)"
Is the fact that Laila and her baby, and other women and their fetuses/babies, were grievously harmed, and that others will be grievously harmed by Directorate of Health's knowledge-obviated, medially & ethically flawed 2104 Recommendation enough justification via FOIA: Section 11: Enhanced access to information (Meirinnsyn): "The administrative agency should allow access if the interest of public access outweighs the need for exemption." But, there it is: "should allow access" is oceans apart from "must allow access."
All this begs the question: Will Directorate of Health be allowed to use Norway's FOIA Exemptions as a shield for willful recklessness, gross negligence and corruption with respect to their knowledge-obviated, medically & ethically flawed 2014 Recommendation? [Note: Yes, sure, consider this is a solicitation for legal scholars to walk the walk by applying their knowledge of case law and legal theory.]
Values of Directorate of Health
Included below is a statement of values from Directorate of Health's webpage: About the Norwegian Directorate of Health.
"Out values are:
The breaches of medical ethics inherent in Directorate of Health's medically & ethically flawed 2014 Recommendation seem too numerous to list. Included below are excerpts from the Norwegian Medical Association (Den norske legeforening) website "Code of Ethics for Doctors" "Adopted by the Representative Body in 1961 and subsequently amended, most recently in 2015." All text in bold font is from "Code of Ethics for Doctors" with comments following in plain text. Needless to say, a medical ethics expert would have a field day and, probably, a nice paper to publish, by analyzing Directorate of Health's medically & ethically flawed 2014 Recommendation. [Note: Yes, that was an invitation to medical ethics experts who talk the talk and walk the walk.] (Source: http://legeforeningen.no/om-legeforeningen/organisasjonen/rad-og-utvalg/organisasjonspolitiske-utvalg/etikk/code-of-ethics-for-doctors-/)
- I. General provisions
§ 2
A doctor shall safeguard the interests and integrity of the individual patient. Patients must be treated with caring and respect. Cooperation with patients should be based on mutual trust and, where possible, on informed consent.
By having had the 18wUSE, Laila's interests, integrity and rights were violated without her prior, informed, voluntary consent when: 1) Laila's combined, fully corroborating, factual LMPD/OTPD/SCID-based GA & EDD and all her other key pregnancy dates were obviated from all medical evidence, 2) Laila and all of her medical professionals were forced to accept an assigned, grossly inaccurate NCFM eSnurra BPD-based EDD and, therefrom, a calculated and correspondingly grossly inaccurate GA, using the equivalent of Naegele's rule, for the duration of Laila's pregnancy, and beyond, all the way to the Medical Birth Registry of Norway (MBRN), thereby demonstrating these critical medical mistake which cause unnecessary, increased medical risks and grievous harms are silently and invisibly written off, unattributed and unreported, as the acceptable collateral damage of Directorate of Health's medically & ethically flawed, intentionally reckless, willfully negligent 2014 Recommendation.
§ 9
In examinations and treatment a doctor shall only employ methods indicated by sound medical practice. Methods which expose the patient to unnecessary risk shall not be employed. If a doctor does not possess the skill a method calls for, he or she shall ensure that the patient receives other competent treatment.
Excluding medical evidence of know and proven medical efficacy in the practice of evidence-obviated medicine is not sound medical practice and it caused Laila and her baby to endure unnecessary, increased risks and grievous harms. Laila's medical professionals had the skills to understand the significance of the triple-combination of: 1) factual last menstrual period date (LMPD), 2) factual ovulation-test-positive date (OTPD) and 3) factual single-coitus-insemination date (SCID) for a combined, fully corroborating, factual LMPD/OTPD/SCID, the trifecta of establishing the beginning of a pregnancy and its temporal frame of reference with LMPD = 0w+0, OTPD = 2w+0 and SCID = 2w+1 for all gestational mathematics. Other that an IVFD, there is nothing more accurate than a combined, fully corroborating, factual LMPD/OTPD/SCID to: 1) provide the best estimate for the beginning of a naturally conceived pregnancy and, therefore, GA & EDD and 2) isolates and eliminate up to 50% of total gestational variance over an entire pregnancy to ensure ovulation variance (follicular phase) is not erroneously allocated by ultrasound dating methods into grossly inaccurate EDD & GA values while masking potential fetal pathology; and, this is precisely what happened and, thereby, Laila and her baby were exposed to unnecessary, increased medical risk which caused unnecessary grievous harms.
A doctor must not use or recommend methods which lack foundations in scientific research or sufficient medical experience. A doctor must not allow him- or herself to be pressed into using medical methods which he or she regards as professionally incorrect.
The practice of evidence-obviated medicine "lacks foundations in scientific research" and lacks, obviously, "sufficient medical experience." Evidence obviated-medicine is always "professionally incorrect." Moreover, Directorate of Health were warned their 2014 Recommendation was medically flawed by Norway's experts in obstetric medicine, fetal medicine, obstetric clinical care and obstetric & fetal medicine research.
When new methods are being tried out, regard for the patient on whom they are being tried shall be the primary concern.
Evidence-obviated medicine was (and is) the new method being tried out. There was no regard nor primary concern for Laila or her baby. All regard and primary concern were reserved for obeying "The Rule" of NCFM eSnurra Group and the Norwegian guidelines. If there had been regard or primary concern for Laila and her baby, at least one of Laila's medical professionals would have compared her factual LMPD/OTPD/SCID-based GA & EDD with the grossly inaccurate BPD-based eSnurra EDD & GA which were forced upon Laila via the 18wUSE when her combined, fully corroborating, factual LMPD/OTPD/SCID and other key pregnancy dates were obviated by government mandate without Laila's prior, informed, voluntary consent.
§ 10
A doctor shall maintain and constantly seek to renew his or her knowledge.
Laila's medical professionals, trainers and administrators need to understand exactly what highly accurate (high sensitivity & specificity) ovulation tests and test systems are, and what a factual ovulation-test-positive date (OTPD) corroborated by a factual LMPD means; and then what it means when a factual single-coitus-insemination date (SCID) completes the triple-combination of LMPD/OTPD/SCID. Also, home pregnancy tests entered the mass market in 1985 and home ovulation tests, in 1989. Home ovulation tests have been available for almost 30-years. And while they are not new, they have continually improved with period/cycle tracking, dual-hormone profiling, monitoring and peak signalling using lab-on-a-chip technologies. And, the truly Orwellian doublethink-required horror of this insidious, systemic problem is, these medical professionals do know what a combined, fully corroborating, factual LMPD/OTPD/SCID means; but they also know there are career consequences, as Laila & Edward were informed, if they do not "follow the rule" of NCFM eSnurra Group and the Norwegian guidelines (i.e., the mandate) that is an insidious, systemic, institutionalized doublethink-required implementation of a government-mandated protocol of evidence-obviated medicine which all must follow, else suffer career consequences. It truly could not be more Orwellian than it already is.
A doctor should according to his or her competence contribute to the development and mediation of medical knowledge.
A woman should not have to explain what a combined, fully corroborating, factual LMPD/OTPD/SCID means in terms of medial evidence to a midwife or doctor charged with providing pregnancy care.
II. Rules governing the relations of doctors with their colleagues and collaborators
§ 5
Public and other debates between colleagues on medical questions and health policy issues must be conducted in an objective manner.
The public debate conducted by Directorate of Health regarding their 2014 Recommendation was an insult to evidence-based medicine, the scientific method and basic reasoning. There was nothing objective about Directorate of Health's decision because their decision process was corrupted from within and by special interests.
III. Marketing and other information concerning medical services
§ 4
A doctor may only use such titles and designations as his or her education and position entitle him to.
Inger Økland, MD was not entitled to use a PhD credential she had not earned and which had never been conferred upon her by NTNU. Also, Økland should never have allowed anyone else to publish a fraudulent PhD credential in her name without making diligent efforts to have a correction published or a retraction issued by the publishers.
He or she may not use titles and designations which may give an erroneous impression of his or her qualifications and work.
Torunn Janbu and Directorate of Health acted with specific intent to deceive by misrepresenting the academic credentials of Økland and her doctoral thesis as being a PhD degree. As a result, Torunn Janbu's intentional misrepresentation of Økland's academic credentials in order to deceive is a breach of medical ethics. Moreover, this misrepresentation of Økland's academic credentials and that of her dr.philos. thesis is a direct breach of Norwegian Medical Association's "Code of Ethics for Doctors" given both Inger Økland and Torunn Janbu are Medical Doctors.
IV. Rules governing the issuing by doctors of medical certificates and other certified documents
§ 1
A medical certificate is a declaration by a doctor concerning a person's state of health. Medical certificates comprise such documents as completed forms for the use of the National Insurance authorities, certificates for various purposes, and statements of expert opinion.
Laila's 18wUSE reports and associated plots/graphs with their NCFM eSnurra assigned EDD & GA were grossly inaccurate and, therefore, completely erroneous and, in the words of NGF "can be directly dangerous." The interpretation or analysis of Laila's 18wUSE was either incompetent or never performed, as the large discrepancy between the NCFM eSnurra BPD-based GA and FL-based GA should have included the MAD-based GA for additional evidence; evidence which also could have been provided by Laila's combined, fully corroborating, factual LMPD/OTPD-based GA & EDD, if it had allowed after it had been obviated by government mandate, but such was not the case. Also, an FGR/malformation of Helen's head was masked by NCFM eSnurra BPD-based EDD & GA because these values were unilaterally interpreted as evidence of GA for an accurately average fetus instead of evidence of both GA and fetal pathology when compared to the NCFM eSnurra FL & MAD-based GA. Also, Laila had been assigned an eSnurra GA & EDD at her first ultrasound exam, in her first-tiemester at LMPD/OTPD/SCID-based GA = 13w+2, which should never have been replaced with the later NCFM eSnurra GA & EDD from Laila's 18wUSE at LMPD/OTPD/SCID-based GA = 19w+4 when fetal metrics and their measurements presented greater random fetal growth velocity variances (i.e., higher standard deviations).
§ 2
A doctor shall not issue a medical certificate if he/she is in doubt as to his/her competence. If a doctor does not find objective grounds for issuing a certificate, a certificate shall not be issued.
In Laila's case, as was the case with Directorate of Health's medically & ethically flawed 2014 Recommendation, there was no medical competence involved prior to Laila's Cesarean section surgery, for which Laila's Cesarean section surgical team were outstanding. [Note: More of a delay on cord clamping would make for a smoother transition to atmosphere respiration; another area of obstetric medicine for which Dr. Hutchon was an innovator.]
§ 3 A doctor shall base his/her certificates on the necessary information and on examinations that are sufficiently extensive for the purpose.
Laila is not aware of any extensive examinations performed prior to her pelvic CT-scan at St Olavs before her Cesarean section surgery delivery.
§ 4
A medical certificate shall convey sufficient information for its purpose and be objective and neutral in its wording. Relevant information must not be withheld or distorted. A certificate shall not contain more information than necessary for its purpose. When medical documents intended for other purposes are attached to medical certificates, special care must be taken to observe professional secrecy.
In Laila's case almost all relevant information was withheld or distorted. At the 18wUSE Laila's LMPD/OTPD/SCID and all of her other key pregnancy dates were obviated, never to be used as medical evidence in the practice of evidence-based medicine.
§ 5
A medical certificate must clearly show to whom it is addressed, its purpose, the doctor's relation to the person concerned, and what the doctor's knowledge concerning the person is based on. Written certificates must be drawn up as separate documents and dated and signed.
None of Laila's ultrasound reports were signed by a doctor. Moreover, there is no evidence that a doctor ever reviewed Laila's 18wUSE report prior to when the grossly inaccurate NCFM eSnurra EDD & GA were assigned to Laila's pregnancy, without Laila's prior, informed, voluntary consent.
American College of Physicians Ethics Manual: Sixth Edition
Included below is an excerpt from the American College of Physicians Ethics Manual: Sixth Edition. The entirety of this manual is available via the links included below. It is interesting to note the ACP Ethics Manual has 43 occurrences of the word "consent" and 16 occurrences of the 2-word sequence "informed consent."
- "Medical and professional ethics often establish positive duties (that is, what one should do) to a greater extent than the law. Current understanding of medical ethics is based on the principles from which positive duties emerge (Table 1). These principles include beneficence (a duty to promote good and act in the best interest of the patient and the health of society) and nonmaleficence (the duty to do no harm to patients). Also included is respect for patient autonomy—the duty to protect and foster a patient's free, uncoerced choices (6). From the principle of respect for autonomy are derived the rules for truth-telling. The relative weight granted to these principles and the conflicts among them often account for the ethical dilemmas that physicians face. Physicians who will be challenged to resolve those dilemmas must have such virtues as compassion, courage, and patience.
In addition, considerations of justice must inform the physician's role as citizen and clinical decisions about resource allocation. The principle of distributive justice requires that we seek to equitably distribute the life-enhancing opportunities afforded by health care. How to accomplish this distribution is the focus of intense debate. More than ever, concerns about justice challenge the traditional role of physician as patient advocate." (Source: American College of Physicians Ethics Manual: Sixth Edition, ACP ETHICS MANUAL, 3 JANUARY 2012 (HTML version) or (PDF version). Lois Snyder, JD; for the American College of Physicians Ethics, Professionalism, and Human Rights Committee (*), Article, Author, and Disclosure Information)
The Biotechnology Act: Excluded the Routine 18-Week Ultrasound Exam
- Ministry of Health and Care Services states: "The Biotechnology Act aims to ensure that biotechnology for medical applications is best utilised for the benefit of a society with a place for everyone."(Source: https://www.regjeringen.no/en/topics/health-and-care/innsikt/biotechnology/id11717/)
- The Biotechnology Council (Bioteknologirådet) states: "The Act relating to the application of biotechnology in medicine is administered by the Ministry of Health and Care Services, and medical use of biotechnology is supervised by the Norwegian Directorate of Health." (Source: Biotechnology Council (Bioteknologirådet) http://www.bioteknologiradet.no/english/norwegian-regulation/)
- "The Act relating to the application of biotechnology in human medicine, etc. About the Biotechnology Act" Source: https://www.regjeringen.no/globalassets/upload/kilde/hod/red/2005/0081/ddd/pdfv/242718-biotechnology_act_master.pdf)
- Act of 5 December 2003 No. 100 relating to the application of biotechnology in human medicine, etc
Cf. earlier Acts of 5 August 1994 No. 56 and 12 June 1987 No. 68
Chapter 1. Purpose and scope
§ 1-1. Purpose of the Act
The purpose of this Act is to ensure that medical applications of biotechnology are utilised for the benefit of everyone in an inclusive society. This shall be done in accordance with the principles of respect for human dignity, human rights and personal integrity and without any discrimination on the basis of genetic constitution, on the basis of the ethical norms that form part of our Western cultural heritage.
§ 1-2. Scope of the Act
This Act applies to the application of biotechnology in human medicine, etc., including medically assisted reproduction, research on embryos and cloning, prenatal diagnosis, postnatal genetic testing, gene therapy, etc. The Act does not apply to research that has no diagnostic or therapeutic consequences for the participant or where data about an individual person are not linked to that person. The provisions of Chapter 3 are excepted from this provision. The Act does not apply to autopsies that come within the scope of Chapter 2 of the Act of 9 February 1973 No. 6 relating to transplantation, hospital autopsies and the donation of bodies and the provisions on expert autopsies, see section 228 of the Criminal Procedure Act of 22 May 1981 No. 25. This Act applies within the realm. The King may by regulations decide that part or all of the Act shall apply in Svalbard and Jan Mayen.
[Note: Regarding the reference to Chapter 3 above: "Chapter 3. Research on embryos and cloning, etc."]
Chapter 4. Prenatal diagnosis
§ 4-1. Definition
For the purpose of this Act, prenatal diagnosis means the examination of fetal cells, a fetus or a pregnant women to obtain information about the genetic constitution of the fetus or to detect or exclude a disease or abnormality of the fetus.
Ultrasound examination that forms part of the ordinary health care offered during pregnancy is not considered to be prenatal diagnosis pursuant to the first paragraph, and therefore does not come within the scope of this Act, with the exception of section 4-5.
§ 4-5. Information on the sex of the fetus before the 12th week of pregnancy
Information on the sex of the fetus before the 12th week of pregnancy resulting from prenatal diagnosis or other examination of the fetus shall only be given if the woman is a carrier of a serious sex-linked disease.
Intentional Recklessness & Willful Negligence
Norway's experts in obstetric medicine, fetal medicine and obstetric clinical care were:
- prevented from reviewing information Directorate of Health claimed they acquired from "comprehensive scientific documentation, assessments by the Norwegian Knowledge Centre for the Health Services and expertise in academic communities in Norway and abroad,"
- excluded from the decision-making process
- ignored when they warned Directorate of Health of the risks and consequences of their medically & ethically flawed 2014 Recommendation, with direct language: "medially flawed"; "critical mistakes my follow"; "muddled investigation process"'; "highly reprehensible" and "can be directly dangerous"
- provide exclusive NCFM eSnurra ultrasound-based estimations of date of delivery (EDD) and, therefrom, calculations of gestational age (GA), using the equivalent of Naegele's rule, in reverse, for all pregnancies in Norway and
- provide exclusive NCFM eSnurra ultrasound-based medical evidence of GA and fetal age, calculated from an NCFM eSnurra BPD-based EDD using the equivalent of Naegele's rule, in reverse, for all abortion adjudications
Directorate of Health's 2014 Recommendation was formulated, decided and implemented in an intentionally substandard fashion, the consequences of which included increased medical risks, critical medical mistakes and grievous medical harms of which Directorate of Health were clearly and explicitly warned by NGF, Bergen Group and others.
The following excerpts articulate the foundation upon which Directorate of Health claimed to have based their medically & ethically flawed 2014 Recommendation.
- "The Directorate’s recommendations are based on comprehensive scientific documentation, assessments by the Norwegian Knowledge Centre for the Health Services and expertise in academic communities in Norway and abroad, including with regard to the statistical methodology." (Source: "One Norwegian national tool for estimating date of delivery and fetal age" Torunn Janbu, Torunn Janbu, MD, PhD, specialist in general and orthopaedic surgery, head of the Department of Hospital Services, Norwegian Directorate of Health. The author has completed the ICMJE form and declares no conflicts of interest., Tidsskr Nor Legeforen nr. 9, 2016; 136, p. 790)
- "Directorate of Health's conclusion is based on several years of dialogue with parts of specialist institutions and Knowledge Center's assessment and experts." (Source: 2015-08-24 Final Minutes of 2015-04-17 April 2015 meeting between the Directorate of Health and the Norwegian Medical Association (Den Norske Legeforening) "about the process behind the recommendation tools for term estimation" 2015-08-24 her - Legeforeningen)
- "It is possible to conduct a systematic search for such studies, but it is unlikely that such a search will lead to relevant findings." ("Det er mulig å gjennomføre et systematisk søk etter slike studier, men det er lite trolig at et slikt søk vil lede til relevante funn.") (Source: Knowledge Centre for Health Services in Public Health, Assessment of the most accurate and research-based tools of gestation and term, Jens Grøgaard, Cecilie (Sommerstad, Forslagsnr: 792, 15.11.2013)
NOKC's Reply to Directorate of Health or "The Fix"
Included below is a translation of the reply NOKC sent to Directorate of Health. NOKC's reply is included in its entirety due to the significance of what NOKC states and, more importantly, what NOKC does not state. URL links have been added to the the Case number and the 5 cited references for ... wait for it ... easy reference.
- Memo To: Health Directorate to Cecilie Sommerstad and Jens Grøgaard
Cc: Torunn Janbu, Postmottak Directorate of Health
Date: 2nd July 2014
Case number: Booking 792, 14/00171
From: Knowledge Center Senior Lecturer: Kjetil Gundro Bruberg Sign .: [Enter Here]
Contact: Liv Merete Reinar, Gro Jamtvedt Sign .: [enter here]
Methods of estimating gestational age and term
Background
Weeks of gestation and term currently have direct implications for the definition of the absolute upper limit for late abortion (21 weeks + 6 days), the lower limit for intensive treatment of extremely premature births, the upper limit of prematurity and overtime pregnancies. In Norway predicted term on the basis of routine ultrasounds in week 17 to 19. Two different ultrasound based methods are in use, respectively, "term wheel" (1) and "esnurra" (2), and the two methods based on different prediction models that do not give identical result (3).
In order to ensure equality across regions and regions, wants health authorities to define the methods and prediction models that will form the basis for the determination of gestational age and futures in Norway. This is why the Knowledge Center in November 2013 received an order from the Directorate of Health in which the Knowledge Center was asked to evaluate the accuracy of various tools to determine gestational age and term.
Assessment of Order
The primary objective of the order from the Directorate of Health is to obtain comparative data for the diagnostic accuracy of "term wheel" versus "esnurra," that here is, the ability to predict straight term and the ability to provide accurate measurements throughout the respective measurement window. Traditional diagnostic validation studies require access to a unified reference standard for the calculation of sensitivity and specificity etc, but for the term estimation, there is no reference standard. It is natural to compare the predicted term with actual date of birth, but since children born both preterm and post-term is not birth a reference standard in the traditional sense. In the absence of reference standard must find other ways to validate the different prediction on.
One possible solution is to compare different prediction models within the framework of controlled studies or time series analysis to see if the selection of prediction affects measurable outcomes. It is possible to carry out a systematic search for such studies, but it is unlikely that such a search will lead to relevant findings.
Another validation strategy is via studies where properties of the current methods are compared directly through measurements in the same population. In her doctoral dissertation from 2012 (4) Inger Økland conducted validation studies of three different prediction models ("Snurra", "Term wheel" and "esnurra"), a work that provides a good basis for assessing the actual differences between those prediction models that are relevant in Norwegian context. Interesting projects are underway, among other things, it created new reference curves for symphysis-fundusmål (5) which is calibrated for use in Norway with "esnurra." Preliminary search does not indicate that, after 2012, published validation studies where "Term wheel" and "esnurra" compared directly.
Conclusion
After an overall assessment, it seems unlikely that a systematic overview carried out in accordance with the Knowledge Center's method handbook will help to highlight the problem beyond what is already known. When the Directorate of Health determines which prediction model should be used to estimate pregnancy length and term, it seems reasonable to base the decision on the findings presented in Inger Økland's doctoral thesis (4).
References
1. Johnsen SL. Rasmussen S, Sollien R, Kiserud T. Fetal age assessment based on femur length that 10 to 25 weeks of gestation, and reference ranges for femur length to head circumference ratios. Acta Obstet Gynecol Scand 2005; 84: 725-733
2. Gjessing HK Grøttum P, Eik-Nes SH. A direct method for ultrasound prediction of day of delivery: a new population based approach. Ultrasound Obstet Gynecol 2007; 30: 19-27
3. Øakland I, Nakling J Gjessing HK Grøttum P, Eik-Nes SH. Advantages of the population-based approach, two pregnancy dating: results from 23020 ultrasound examinations. Ultrasound Obstet Gynecol 2012; 39: 563-568
4. Økland I. Bias in second-trimester ultrasound dating related to prediction models and fetal measurements. Doctoral thesis, NTNU: 2012 Pay AS, Frøen JF, Staff AC, Jacobsson B, Gjessing HK. A new population-based reference curve for symphysis fundus height. Acta Obstet Gynecol Scand 2013; 92: 925-3
5. Pay AS, Frøen JF, Staff AC, Jacobsson B, Gjessing HK. A new population-based reference curve for symphysis-fundus height. Acta Obstet Gynecol Scand 2013; 92: 925-33
Norway's FOIA Exemptions: A Potential Shield for Corruption & Willful Negligence?
The many exemptions in Norway's Freedom of Information Act (offentleglova) can be used to shield Directorate of Health's: unethical behavior, breach of medical ethics, professional misconduct, conflict of interest, acting beyond competence, actively ignoring warnings of the risks and consequences from Norway's medical experts, thereby, engaging in intentional recklessness, willful negligence, and, possibly, criminal activity that served to corrupt Directorate of Health's:
- decision-making processes,
- decision (or lack thereof via "the fix") and
- decision implementation protocol & process
- increased medical risks, critical medical mistakes and grievous medical harms to some of Norway's women and their fetuses/babies and
- Norway's medical professionals (i.e., midwives, nurses, doctors & specialists) are forced to practice evidence-obviated medicine instead of evidence-based medicine with respect obstetric medicine, fetal medicine and obstetric clinical care.
- conscious disregard of their knowledge of identified, known risks and consequences; published risks and consequences which had been made explicit by Norway's medical experts in easy-to-understand language:
- NGF: "reprehensible" and "can be directly dangerous" and
- Bergen Group: "medically flawed" and "critical mistakes may follow" and "muddled investigation process"
- conscious disregard of the identified, known risks and consequences; consequences proven to include increased medical risks, critical medical mistakes and grievous medical harms.
FOIA Exemption: Internal Documents
The interest of public access to information, especially regarding the formulation and decision-making processes of national healthcare policy and associated implementation protocols, must outweigh the need for FOIA exemption. For reference, Norway's Freedom of Information Act (FOIA) is known as Lov om rett til innsyn i dokument i offentleg verksemd (offentleglova) which is also known as: "Act of 19 May 2006 No. 16 relating to the right of access to documents held by public authorities and public undertakings (short title: Freedom of Information Act (offentleglova)"
- English: https://lovdata.no/dokument/NLE/lov/2006-05-19-16: Act relating to the right of access to documents held by public authorities and public undertakings (Freedom of Information Act)
- Norwegian: LOV-2006-05-19-16: Lov om rett til innsyn i dokument i offentleg verksemd (offentleglova) [official version]
- "The definition of “internal documents” was narrowed in the 2006 FOIA.34 As for external documents, the FOIA allows an authority to exempt from public access documents that it has obtained from subordinate bodies for “its internal preparation of a case”, that is for pre-decisional and deliberative purposes. There is a corresponding exception for information that one government department obtains from another department, as well as an exception for external advice that the agency has received about how it should handle a case." (Source: THE NORWEGIAN FREEDOM OF INFORMATION ACT – A NOT SO TRANSPARENT ACT? by Iris NGUYÊN-DUY, Postdoctoral research fellow, Faculty of Law, University of Oslo, URL link)
- FOIA: Section 11
Enhanced access to information
"Where there is occasion to exempt information from access, an administrative agency shall nonetheless consider allowing full or partial access. The administrative agency should allow access if the interest of public access outweighs the need for exemption."
FOIA: Section 14
Documents drawn up for an administrative agency’s internal preparation of a case (internal documents)
An administrative agency may exempt from access any document which it has drawn up for its internal preparation of a case.
The first paragraph does not apply to:
(a) any document or part of a document containing the final decision of the administrative agency in a case,
(b) general guidelines for the administrative agency’s case processing,
(c) reasons for proposals that have been decided by the King in Council, and
(d) brief descriptions of the content of documents and the like, but not if such a description reproduces internal assessments.
The King may make regulations providing that exemptions may not be made pursuant to the first paragraph of this section in respect of specific documents in specific state or state-owned agencies.
FOIA: Section 15
Documents obtained externally for internal preparation of a case
Where it is necessary in order to ensure proper internal decision processes, an administrative agency may exempt from access any document that the agency has obtained from a subordinate agency for use in its internal preparation of a case. The same applies to documents which a ministry has obtained from another ministry for use in its internal preparation of a case.
Moreover, exemptions may be made in respect of parts of any document containing advice on and assessments of how an administrative agency should stand on a case, and which the agency has obtained for use in its internal preparation of the case, where this is required in the interest of satisfactory protection of the government’s interests in that case. The exemptions in this section apply correspondingly to documents concerning the acquisition of a document as mentioned in the first and second paragraphs, and to notices of and minutes from meetings between a superior and subordinate agency, between ministries and between an administrative agency and any person who gives advice or assessments as mentioned in the second paragraph.
This section does not apply to documents obtained as part of the general procedure of consultation on a matter.
(Source: Act of 19 May 2006 No. 16 relating to the right of access to documents held by public authorities and public undertakings (Freedom of Information Act), with later amendments up to and including Act of 28 April 2017 No. 22.)
Is the fact that Laila and her baby, and other women and their fetuses/babies, were grievously harmed, and that others will be grievously harmed by Directorate of Health's knowledge-obviated, medially & ethically flawed 2104 Recommendation enough justification via FOIA: Section 11: Enhanced access to information (Meirinnsyn): "The administrative agency should allow access if the interest of public access outweighs the need for exemption." But, there it is: "should allow access" is oceans apart from "must allow access."
All this begs the question: Will Directorate of Health be allowed to use Norway's FOIA Exemptions as a shield for willful recklessness, gross negligence and corruption with respect to their knowledge-obviated, medically & ethically flawed 2014 Recommendation? [Note: Yes, sure, consider this is a solicitation for legal scholars to walk the walk by applying their knowledge of case law and legal theory.]
Values of Directorate of Health
Included below is a statement of values from Directorate of Health's webpage: About the Norwegian Directorate of Health.
"Out values are:
- Professionalism: The Directorate's work is knowledge-based and relies on exercising sound professional judgment
- Openness: The Directorate's work is characterised by clarity, transparency, open processes and clear communication
- Cooperation: The Directorate's work entails a comprehensive, interdisciplinary approach and extensive dialogue"